5.17 Hyphal fusions and mycelial interconnections
The hyphal tip we have just described is capable of forward extension for an indefinite period, or at least for as long as nutrients are available and environmental conditions remain amenable. But the hyphal tip has another characteristic capability: it can fuse with another hypha. Hyphal fusion (also called anastomosis) is a ubiquitous phenomenon in filamentous fungi, serving many important functions at different stages in the life cycle.
At the very earliest stages of the vegetative phase, when the spore is germinating, fusions can occur between the germ tubes of clustered spores as they germinate. Subsequently, in the interior of the maturing vegetative colony, numerous hyphal fusions occur to convert the radially-oriented main hyphae of the mycelium into an interconnected network. This is essential for intra-hyphal communication within the colony, particularly translocation of water and nutrients, and maintenance of general homeostasis (Trinci et al., 1994, 2001). Also, entry into the sexual cycle usually involves the fusions between hyphae of the two parents, and then maintenance of the dikaryotic state, the prelude to karyogamy, also requires anastomoses, but this time in specialised structures called croziers (in the ascogenous hyphae of Ascomycota) or clamp connections (associated with the vegetative dikaryotic hyphae of Basidiomycota). Hyphal fusion is also involved in the formation of the multicellular tissues found in fruit bodies and other sporing structures.
We will discuss later the mechanisms that exert control over hyphal fusion by defining vegetative and sexual compatibility (see Section 7.5 and Chapter 8); here we will outline the cell biology of hyphal fusion, which has been a major research focus for several years (Fig. 12; see Hickey et al., 2002; Glass et al., 2004; Wright et al., 2007; Read et al., 2009, 2010; Read & Roca, 2013; Raudaskoski, 2015; Fleißner & Serrano, 2016).
Three types of pre-contact behaviour have been described:
- a hyphal tip induces a branch and they subsequently fuse (shown in Fig. 12B);
- two hyphal tips approach each other and subsequently fuse;
- a hyphal tip approaches the side of another existing hypha and there is a tip-to-side fusion (shown in Fig. 12C).
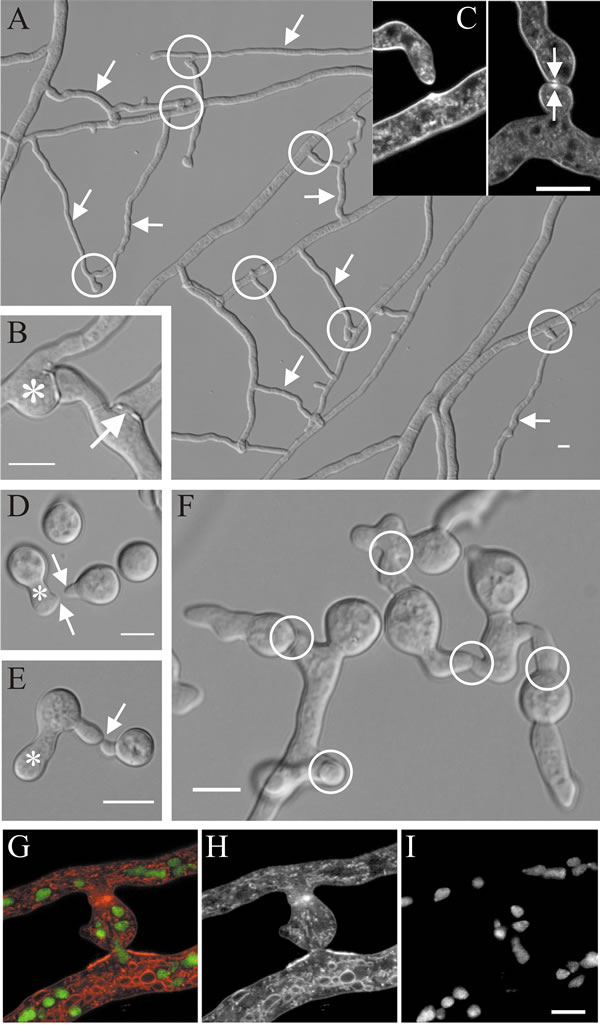 |
| Fig. 12. Hyphal fusions in Neurospora crassa. A, shows a region about 1 to 2 cm behind the leading edge of a mature mycelium in which specialised fusion hyphae (some are arrowed) fuse with other hyphae to establish a colony network (some of the fusion connections are circled). B, detail of a hyphal fusion connection showing isotropic swelling upon contact (asterisk) and a fusion pore (arrow). C, confocal images of hyphal homing and fusion in Neurospora crassa: the image at left shows a hyphal tip growing towards a hyphal peg (pre-contact stage), with a brightly fluorescent Spitzenkörper evident in both the hyphal tip and emerging peg; the right hand image shows the post-contact stage in which the adherent hyphal tips have swollen but their fluorescent Spitzenkörpers (arrowed) have persisted even though hyphal extension has ceased. Self-fusion in Neurospora crassa occurs during early as well as late stages of colony development. D, after an initial phase of isotropic expansion, conidia of N. crassa polarise leading to the outgrowth of germ tubes (asterisk) and conidial anastomosis tubes (called ‘CATs’) that can arise directly from conidia or from germ tubes (arrows); in this specimen the tip of the right hand CAT seems to have induced the formation of the CAT from the germ tube. E, CATs chemotropically attract (since these are conidia from the same mycelium this is an autotropism) and become attached to each other (arrow indicates site of contact); when they come into contact, tip growth is arrested and a fusion pore is formed. Each germinating conidium can interact with several neighbours (F), thereby creating an interconnected network (CAT connections are circled in F) of newly-germinated conidia (germlings). G-I, confocal images showing fused hyphal branches of Neurospora crassa through which nuclei are flowing stained with specific fluorescent probes: G is the combined image (colour-coded); H shows fluorescence of membrane-specific labelling alone; I shows only the fluorescence of nuclei specifically labelled with histone-specific staining. Scale bars = 10 µm. Modified from Hickey et al., 2002; Glass et al., 2004; Read et al., 2009, using graphic files kindly provided by Prof. N. D. Read, Institute of Cell Biology, University of Edinburgh. Images reproduced with permission of Elsevier. |
The Spitzenkörper is closely involved in the process of hyphal fusion, which have been characterised into nine stages (Glass et al., 2004) as follows:
- Stage 1: a hyphal tip competent to initiate fusion secretes an unknown diffusible, extracellular signal which induces Spitzenkörper formation in the hypha it is approaching; it is presumed that the second hypha secretes a corresponding signal;
- Stages 2 and 3: these events result in two fusion-competent hyphal tips each secreting diffusible, extracellular chemotropic signals that regulate Spitzenkörper behaviour so that the hyphal tips grow towards each other (Fig. 13);
- Stage 4: cell walls of the approaching hyphal tips make contact, apical extension ceases, but both Spitzenkörpers persist;
- Stage 5: adhesive material is secreted at the hyphal tips;
- Stage 6: polarised apical extension is converted to ‘all-over’ isotropic growth, which results in swelling of the adherent hyphal tips;
- Stage 7: cell walls and adhesive material at the point of contact are dissolved, bringing the two plasma membranes into contact;
- Stage 8: plasma membranes of the two hyphal tips fuse, creating a pore with which the Spitzenkörper stays associated as the pore begins to widen and cytoplasm starts to flow between the now connected hyphae;
- Stage 9: the pore widens, Spitzenkörper disappears, organelles, including nuclei, vacuoles and mitochondria, can flow between the fused hyphae, though the flow may be regulated.
The principal lesson to take away from this brief description of hyphal fusion can be expressed as: what the Spitzenkörper has put together, the Spitzenkörper can take apart.
For anastomosis to occur between two hyphal tips, two separate cell walls must be disassembled and two separate plasma membranes stitched together around a newly-formed hypha-wide pore, and all of this must be done without breaching the integrity of walls and membranes that are under osmotic hydraulic stress. Add the need for signalling and targeting systems to ensure that fusions occur at the right place and right time, and you can see that the phrase ‘hyphal fusion’ covers an extensive range of cell biological processes that are under delicate control.
It will be some time before this entire process is understood, but a model of self-signalling between hyphal tips of the same mycelium is emerging (Read et al., 2009, 2010). The model (summarised in Fig. 13) imagines that approaching tips alternately signal each other and after receiving a signal a tip grows along the gradient of the signalling molecule (autotropism). In the first half of the signal exchange one of two approaching tips releases the chemoattractant signal while the other responds to the signal by adjusting its growth direction towards the signaller. In the second half of the conversation, the roles are reversed and the second tip releases signal to give the first one a target to adjust its growth direction. The exchange will be repeated until the two tips come into contact.
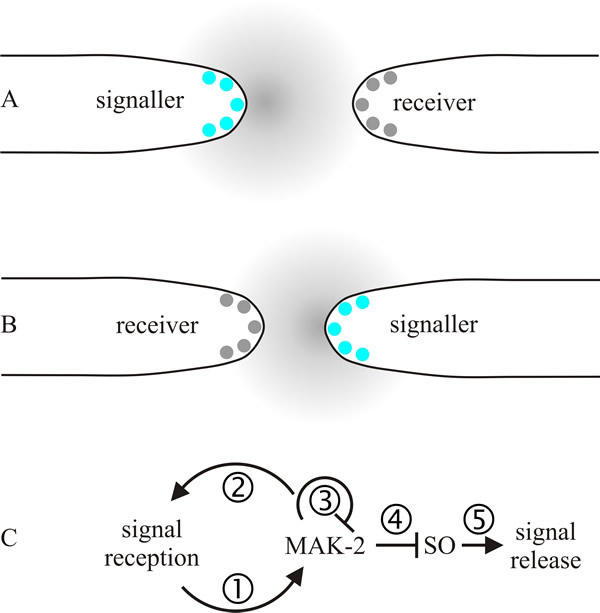 |
| Fig. 13. A model of self-signalling between hyphal tips of the same mycelium. The model imagines that approaching tips periodically and alternately signal each other (called a ping-pong mechanism) and after receiving a signal a tip grows along the gradient of the signalling molecule (autotropism). A shows the first half of the signal exchange; the tip on the left releases the chemoattractant signal while the one on the right responds to the signal by adjusting its growth direction towards the signaller. In B, the second half of the conversation, the roles are reversed and the right hand tip releases signal so that the tip on the left can adjust its growth direction. The exchange will be repeated until the two tips come into contact. Panel C puts some detail on this proposed mechanism of signalling: (1) a chemoattractant-receptor complex induces local recruitment and activation of a mitogen-activated protein (MAP) kinase pathway (here designated MAK-2); (2) positive feedback inherent in MAP kinase amplifies the received signal; (3) as its local concentration increases, one of the MAK-2 proteins down regulates the MAP kinase protein complex; (4) decrease in MAK-2 derepresses the protein SO (specified by a Neurospora crassa gene locus called soft, which encodes a protein containing a domain involved in mediating protein-protein interactions via proline-rich regions). The soft gene is required for vegetative hyphal fusion. Accumulation of SO leads to the formation of SO-containing protein complexes at the tip; (5) a burst of SO-stimulated chemoattractant release occurs. Modified and redrawn from Read et al., 2009. |
The proposed mechanism of signalling, called a ping-pong mechanism (Fig. 13C), involves periodic and alternate signalling between two approaching hyphal tips. An elaborate signalling network has been identified that controls communication, attraction, and merger of fusing hyphae. The network contains many well-conserved elements, including MAP kinases, systems that generate reactive oxygen, Ca2+-binding regulators, cell polarity factors, and proteins homologous to the mammalian ‘striatin-interacting phosphatase and kinase’ (STRIPAK) complex. The mammalian striatin family consists of three proteins that have no intrinsic catalytic activity but serve as scaffolding proteins, which organise many varied, and large, signalling complexes that contribute to a variety of cellular processes. In mammals they are involved in cell signalling, cell cycle control, apoptosis, vesicular trafficking, Golgi assembly, cell polarity, cell migration, neural and vascular development, and cardiac function. They have been connected to clinical conditions, including cardiac disease, diabetes, autism, and cerebral malformations (Hwang & Pallas, 2014). It is no surprise that a system that plays so many crucial roles in a sister clade of organisms also has many essential roles in fungi. STRIPAK homologues are involved in cell cycle arrest and mating in budding yeast, signalling transition from mitosis to cytokinesis in Schizosaccharomyces pombe, and the cell cycle, septal dissolution, nuclear migration, clamp cell development and fusion, and hyphal fusions in filamentous ascomycetes and basidiomycetes (Raudaskoski, 2015; Fleißner & Serrano, 2016; Serrano et al., 2017).
Updated September, 2020
