5.1 Mechanisms of mycelial growth
Polarised growth of fungal hyphae is achieved by restricting extension to the hyphal apex. The cell wall at the hyphal tip has viscoelastic properties [definition] and yields to the internal turgor pressure within the hypha. Further behind the tip the wall is rigidified and resistant to the turgor forces resulting from the osmotic flow of water into the hypha. Turgor pressure generated within the hypha therefore acts as the driving force for hyphal extension.
Hyphal extension at the apex requires synthesis and insertion of new wall material and new membranes in a way that does not weaken the tip. This highly organised process is supported by the continuous flow of vesicles generated within the cytoplasm behind the tip, and is co-ordinated with the growth and replication of all the other cytoplasmic organelles and their migration towards the extending apex. In this Chapter we will be describing all these individual processes, and in Chapter 6 this mosaic of processes will be assembled into what we hope will be a full picture of hyphal tip growth. What is important at this point is to recognise how much of the detailed cell biology of filamentous fungi is adapted, devoted and committed to forward thrusting of the hyphal apex; that is, hyphal extension growth. Making this the supreme characteristic of filamentous fungi that sets them off from the other crown eukaryotes, animals and plants.
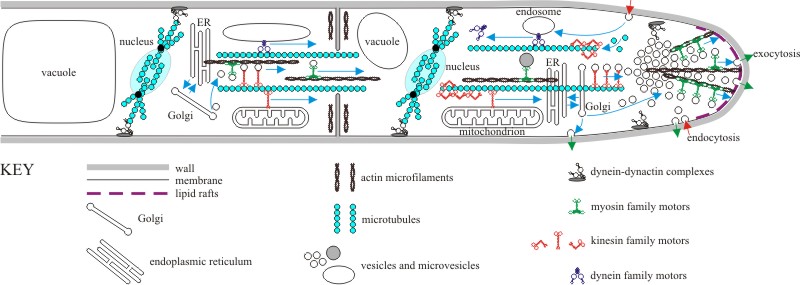 |
|
| Cartoon representation of an overall molecular model of hyphal growth. The key feature of hyphal apical growth is rapid movement towards the apex of all the materials needed to create new wall, new membranes and new cytoplasmic components. Most of these materials are exported in vesicles by the endoplasmic reticulum (ER) and Golgi organelles, the vesicles being delivered to the apical vesicle cluster (called the Spitzenkörper; see Section 5.15 and Fig. 5.5) along microtubules powered by motor proteins of the kinesin and dynein families (see Section 5.12 and Fig. 5.8). The Spitzenkörper organises the final distribution of microvesicles along actin microfilaments to the plasma membrane at the extending tip. Vesicle fusion with the membrane is enabled by t-SNARE and v-SNARE proteins. Sterol-rich ‘lipid rafts’ at the hyphal tip could provide domains for apical proteins like signalling and binding complexes and might facilitate endocytosis. Endocytosis at the hyphal tip is dependent upon actin patches where myosin-1 polymerises actin into filaments that take the endocytotic vesicles away from the membrane. The extreme apex of hyphal tips undergoes extensive exocytosis, which is mainly devoted to synthesis of wall polymers outside the membrane and wall construction and maturation (Sections 6.3 and 6.4 and Figs 6.2 and 6.3). Endocytosis features in the flanking regions of the hyphal tip, and this both recycles membrane components (originally delivered as exocytotic vesicles) and imports nutrients; both of which are transported to the endomembrane system for sorting and appropriate use (see Sections 5.10 and 5.12 and Fig. 5.5). This figure also shows that (potentially many) subterminal hyphal cells contribute to the apical migration of resources; (streams of) vesicles, (trains of rapidly moving) vacuoles (Section 5.12) and mitochondria are all transported towards the apex and this transport extends through hyphal septa. Also note that the position of nuclear division spindles is probably specified by interaction between astral microtubules and membrane-bound dynein-dynactin complexes (Figs 5.10 and 5.11), and septal positioning is associated with rings of actin microfilaments (Section 5.17). Remember: this IS a cartoon, no attempt is made to portray relative scale or relative timing (some structures, like division spindles) are more transient than others (like the Spitzenkörper). Also, everything happens, quickly; in the text (Sections 5.12 and 5.15) we show that 38,000 vesicles have to fuse with the apical membrane each minute (that’s over 600 every second) to support extension of each hyphal tip of Neurospora crassa when it is growing at its maximum rate. See text of Chapters 5 and 6 for complete explanation, and refer to Steinberg (2007) and Rittenour, Si & Harris (2009). |
Updated July, 2019
