2. Stem and cap morphogenesis in Coprinopsis fruit bodies
The stems of Coprinopsis fruit bodies have a totally different metabolism to the caps they support. Stem development and maturation also depend on cell enlargement in the mostly vertically-arranged stem hyphae. Cytologically, this is biphasic, with an initial slow increase in volume being attributable to an increase in hyphal cell diameter in many of the hyphae of the outer regions of the stem, to give an undifferentiated dikaryotic central region and a differentiated cortex made up of multinucleate inflated cells interspersed with entwining dikaryotic hyphae. In the second phase of stem growth these inflated cells increase considerably and rapidly in length in a manner which is co-ordinated with cell inflation events in the maturing cap (Hammad, Ji, Watling & Moore, 1993a; Hammad, Watling & Moore, 1993b; see discussion below). This stem growth depends on reallocation of cellular metabolites, but no single metabolite can be identified as being of prime importance although a few simple sugars do appear to be correlated with the osmoregulatory activity connected with the large scale uptake of water involved in stem elongation.
During stem development glycogen deposition is at first restricted to cells forming a cup-shaped mass in the stem base, but very early in primordium development this deposit is depleted and glycogen accumulates in the gills, particularly the subhymenium, where it probably serves as a reserve material for the later stages of cap development. The distribution of cytochemically detectable protein differs from that of glycogen in that protein initially accumulates in the upper regions of the primordial stem and in the gill hymenium of the cap. The stem showed no conspicuous change in its content of protein during development although in the cap the fraction of the dry weight represented by protein increased substantially (Ewaze, Moore & Stewart, 1978; Moore, Elhiti & Butler, 1979). Change in the ratio of fresh weight to dry weight in the stem as it elongates is such that while reducing sugars will account for about 6% of the dry weight of a (young) primordial stem, they will account for at least 12% of the dry weight of the mature Stage 5 stem.
Together, trehalose and alcohol-soluble reducing sugars represent about 30% of the dry weight of the mature stem. In Agaricus mannitol can amount to as much as 50% of the fruit body dry weight (Hammond & Nichols, 1976) but it does not seem to have any role in Coprinopsis cinerea as total polyols never exceed 6% of the Coprinopsis dry weight and declined in quantity as the fruit body developed. As a matter of interest, we later used gas-liquid chromatography (GLC) on the various tissues of the shiitake mushroom, Lentinula edodes, comparing this fungus with Agaricus bisporus and Coprinopsis cinerea (Tan & Moore, 1994). We found that mycelia of L. edodes had a low level of mannitol (about 1% on a dry weight basis) but high concentrations of mannitol (20-30% of dry weight) in the shiitake mushroom, both stem and cap. The highest level of mannitol was observed in the cap of A. bisporus (close to the 50% of dry weight previously reported). In this study, no mannitol was detectable in the cap of C. cinerea (Tan & Moore, 1994).
Getting back to Coprinopsis, we have good reason to suppose that simple carbohydrates serve an osmoregulatory function in the developing stem of C. cinerea, but no single sugar can be identified as being of prime importance; it seems more likely that a diverse metabolism contributes several carbohydrate components to an osmoregulatory cocktail. It must be emphasised that these considerations refer to what might be called the adjustable osmoticum. Because of the low dry weight to fresh weight ratio (from about 1:10 in the youngest fruit bodies to 1:20 in mature fruit bodies; see Table 2 in Moore, Elhiti & Butler, 1979) high molecular weight organic compounds like sugars cannot contribute greatly to the solute concentrations of the cell contents. Consequently, the bulk of the osmotic pressure of the cells of the stem must be contributed by compounds of low molecular weight and it is probable that inorganic ions (particularly potassium) make a very important contribution, as they do in plants. However, although the sugar content accounts for only a small part of the overall osmolarity of the cell, it does represents a portion which is readily adjusted by metabolism of cell constituents; a property which is not possessed by the inorganic components of the cell.
Aspects of cap development. We concluded that cap development is regulated by a metabolism quite distinct from those of either the parental mycelium or the stem. As carbohydrate nutrients in the medium approach exhaustion suppression of mycelial morphogenesis into hyphal aggregates is lifted, but the subsequent switch from the vegetative pathway of sclerotium development to fruit body formation is normally dependent on illumination with low intensity blue light. Growth then becomes polarised to form a presumptive cap in the upper hemisphere of the multicellular initial aggregate and glycogen is mobilised from the surrounding mycelium to accumulate in the lower hemisphere of that initial (the presumptive base of the stem). Receipt of a second illumination inhibits further growth of the stem base and causes glycogen reserves to be translocated to the cap. In the cap of the later primordium stage the main tissues of the cap are demarcated.
The blue light photoreceptor has been identified by Terashima, Yuki, Muraguchi, Akiyama & Kamada (2005) as the product of a gene called dst1. Mutants of this gene produce a fruit body phenotype only seen in the wild type when fruit bodies are kept in continuous darkness, when primordia do not mature but instead the upper part of the primordium remains rudimentary while the basal part elongates, producing a configuration called ‘dark stipe’ (hence the gene symbol dst). The dst1 gene was cloned and the sequence found to predict a protein of 1175 amino acids that contains several structural domains, at least one of which was similar to the flavin binding sites of known blue light receptors, suggesting that the Dst1 gene specifies a blue-light receptor of Coprinopsis cinerea. In a later study, Kuratani, Tanaka, Terashima, Muraguchi, Nakazawa, Nakahori & Kamada (2010) showed that their dst2 mutant, which causes a similar ‘blind’ mutant phenotype (but also affects asexual spore production) encodes a protein with a flavin-adenine dinucleotide (FAD) binding domain; such proteins are known to be component parts of blue light sensors. Dst2 may play a role in perceiving blue light with Dst1 being responsible for communication of the photoreception signal.
Kertesz-Chaloupková, Walser, Granado, Aebi & Kües (1998) had earlier shown that blue light overrides repression of asexual sporulation (that is, production of oidia) by mating type genes in Coprinopsis cinerea. They found that mycelia with mutations in both mating type loci (Amut Bmut homokaryons) also produce copious oidia but only when exposed to blue light (Srivilai, Loutchanwoot & Sukha, 2009). Other developmental events influenced by the A mating type factor also depended on blue light. In monokaryons transformed with compatible A mating type genes (A-activated transformants) production of oidia is repressed in the dark and induced by blue light. Five of six monokaryons tested following transformation with A genes showed induced production of hyphal knots and sclerotia in the dark, and at least three strains showed enhanced chlamydospore production in the dark. Continuous incubation under blue light inhibited formation of hyphal knots, sclerotia and chlamydospores in both competent monokaryons and in A-activated transformants. On artificial medium and on a 12 h light/12 h dark regime, one of the A-activated monokaryon transformants formed fruit-body primordia that were arrested in development before karyogamy (Kües, Granado, Hermann, Boulianne, Kertesz-Chaloupková & Aebi, 1998). These authors suggest that A mating type genes and blue light perception are involved in control of all major differentiation processes in Coprinopsis.
The subhymenium, which lies beneath the tissue layer (hymenium) from which the spores arise, is an open tissue of interwoven hyphae. Cells of the subhymenium at this stage contain large accumulations of glycogen. The hymenium is formed from dikaryotic branches of the subhymenial hyphae to become an organised layer of hyphal tips of determinate growth. The phrase 'of determinate growth' means that this entire community of closely appressed hyphal tips stop growing after a short while, coming to a halt in concert so that they are all of very similar length and together they form a very smooth layer (which is the embryonic hymenium of the gill). The vast majority of these hyphal tips will form basidia (in this young state they are called probasidia), a minority, which are the only ones that continue to grow in size, will form cystidia. But the hymenium is completed by branches which arise from the base of the probasidia (two to five branches from each) and insert their apices into the embryonic hymenium; these are the young paraphyses.
Subsequently, although the subhymenial hyphae remain hyphal, the hymenial paraphyses become greatly inflated. The paraphyses come to form a pavement of interlocked cells pressed closely against each other, all containing a large central vacuole. Basidia are only slightly enlarged and contain dense cytoplasm with few vacuoles; they contrast with the much extended and inflated cystidia. Despite their inflation, all three cell types still show continuity with the subhymenial hyphae (Moore, Elhiti & Butler, 1979). During cap expansion the major inflationary force is enlargement of the paraphyses but as the gill lamellae are removed by autodigestion further cell inflation in the outer flesh of the cap enables it to assume a supportive role. In the earlier stages declining glycogen content of the cap correlates with increasing activity of NADP-linked glutamate dehydrogenase. As discussed above, cap expansion is driven osmotically and at least some of the osmotically active materials are synthesised through the urea cycle. At first this activity could be supported by glycogenolysis and amination but autodigestion of spent gill tissues may provide substrates later in the maturation process. However, there is more to the regulatory circuit surrounding glycogen and NADP-linked glutamate dehydrogenase than has been mentioned so far.
Firstly, we found that intracellular glycogen stimulates fruiting in Coprinopsis cinerea (Brunt & Moore, 1989). This study involved characterising the phenomenon known as renewed fruiting, which is a regeneration phenomenon in which fragments of fruit bodies explanted onto agar media in Petri dishes quickly produce a new crop of fruit bodies. Fruiting directly from the explanted fragment was initiated 2 to 3 days before fruit-body initials were visible on the outgrown mycelium. The yield of fruit bodies on the outgrown mycelium was not correlated with the glycogen content of the culture inoculum (the original fruit body fragment), whereas the yield of fruits arising directly on the initial inoculum showed a positive correlation (coefficient of 0.88) with the fragment’s glycogen content. Supplementation of the medium with commercial rabbit liver glycogen had no such effect (Brunt & Moore, 1989). Intracellular glycogen features prominently in various aspects of growth and development in Coprinopsis and these data suggest that the molecule may act as something more than a mere carbohydrate store.
A little earlier we had demonstrated that addition of cyclic-AMP to specific sites in plate cultures of a mature dikaryotic colony of Coprinopsis cinerea caused a two-fold increase in the amount of glycogen over the whole culture within 19 hours (Kuhad, Rosin & Moore, 1987). There was no evidence for non-random distribution of glycogen around the site of administration of the cAMP. In the normally developing fruit body levels of endogenous cAMP were considerably elevated in fruit body initials and remained unusually high in the fruit body until very late stages of development, when they declined to about the concentration range observed in mycelium. These cAMP accumulations were positively correlated with localised accumulations of glycogen within the fruit body. Published data from in vitro analyses suggest that cAMP may inhibit glycogen synthesis and activate glycogen breakdown in Coprinopsis. This would be consistent with the interpretation that cAMP could be involved specifically in the bulk translocation of glycogen, i.e. cAMP promotes glycogen breakdown in order to mobilise carbohydrate for transport, rather than immediate catabolism. Bearing in mind the effect of 2-deoxy-D-glucose described above (View now) which implies that when glycolysis is stalled and regeneration of ATP is prevented, ADP is utilised for phosphorylation and cAMP may accumulate. So the model developed is that when high levels of synthetic activity deplete supplies of glycolytic metabolites, accumulated cAMP acts as a ‘come hither’ signal to draw supplies of glycogen into the region of need (Kuhad, Rosin & Moore, 1987).
Ji & Moore (1993) showed that the major increase in glycogen concentration in the fruit body cap was initiated as karyogamy became evident in normally developing fruit bodies and the onset of large-scale utilisation of accumulated glycogen was a postmeiotic event. The temporal pattern of glycogen accumulation/degradation was very variable among 23 different fruit body caps when judged on a common developmental time scale (Table 2). This indicates that glycogen content was not closely associated with meiosis or basidiospore formation even though glycogen degradation usually started from about the onset of the latter. Glycogen phosphorylase was the only glycogen degradation enzyme found in cap tissues. Glycogen synthase activity could be demonstrated (and increased with time) even during the phase of most rapid glycogen degradation. Analysis of glycogen contents during in vitro gill development and in sporeless, stem elongationless and cap expansionless mutants suggested that glycogen degradation was not specific for any one of these processes. Nutrient translocation occurred mainly in the 'stem to cap' direction and glycogen influx to the cap continued into the phase of glycogen degradation in the cap. The overall conclusion is that glycogen acts as a carbohydrate supply which cannot be linked exclusively, or even predominantly, with any one of the several processes which occur as the fruit body matures but rather serves any or all of those processes as required. Glycogen is a generalised, easily translocated, but above all transient store of carbohydrate at all stages in the life cycle of Coprinopsis cinerea (Ji & Moore, 1993).
Attempts to use cytochemical tests to localise glutamate dehydrogenase enzymes in live tissue prepared for light and electron microscopy were extremely successful. The NAD- and NADP-linked glutamate dehydrogenases were detected in frozen sections of fruit body tissues by means of tetrazolium salt (the clear chromogen is turned into a blue stain by the dehydrogenase reaction). Material at all stages of development was examined. From the earliest stages NAD-glutamate dehydrogenase was uniformly distributed in the basidial cells of the hymenium, and as the fruit body developed an increasing proportion of cells of the subhymenium came to express this enzyme activity. The NADP-glutamate dehydrogenase (NADP-GDH) was initially limited to basidial cells in isolated patches of the hymenium (Fig. 19). As development proceeded the proportion of basidia exhibiting NADP-GDH activity increased, as did the proportion of subhymenial cells showing this enzyme activity. No NADP-GDH activity was detectable in sections of stem tissue (Elhiti, Butler & Moore, 1979).
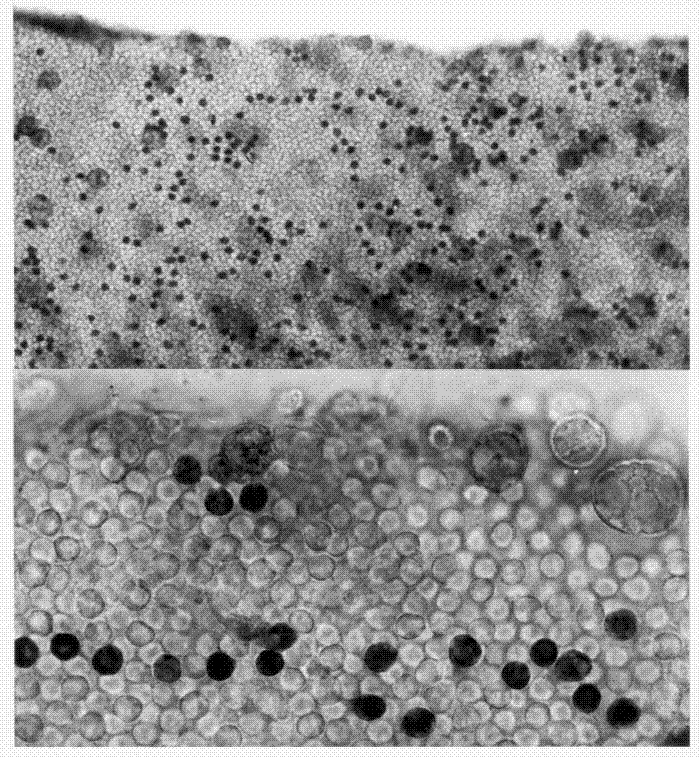 |
| Fig. 19. Application of cytochemical techniques for detection of NADP-GDH activity in situ in live tissues shows that at the earliest stages of fruit body development NADP-GDH is found in a few scattered basidia. These photomicrographs are surface views of the hymenium of extremely young gills excised from fruit body primordia. The tissue was treated so that cells containing NADP-GDH stain darkly (using the tetrazolium reaction). Apart from the few large cystidia, all the cells visible in these tissues are young basidia. Clearly, only a minority contain the enzyme at this early stage of development. Those that do stain may emerge as branches from the same tramal hyphae, because they seem often to fall into lines in the hymenium (bottom image). |
We used electron microscopic cytochemistry to show derepression of NADP-linked glutamate dehydrogenase activity following transfer of mycelium from nitrogen-rich to nitrogen-free medium [experiment described above]. After tests of a number of techniques, a copper ferrocyanide method was adopted as a routine method to visualise dehydrogenase enzyme activity for observation with the transmission electron microscope. In this method, ferricyanide is used as an electron acceptor, and is converted on reduction into insoluble electron-opaque cupric ferrocyanide. Tissue was fixed in buffered formaldehyde solution for 20 to 25 minutes at 4°C, washed in 0.1 M phosphate buffer, pH 7.2 with sucrose, and incubated in copper ferricyanide medium for 90 min at 37°C. After several washes in buffer with sucrose, the materials were post-fixed in buffered 2% osmium tetroxide for 1 hour at 40°C, dehydrated in ethanol and embedded in epoxy resin for preparation of ultrathin sections (Elhiti, Moore & Butler, 1986). In the tissue sections the NADP-linked glutamate dehydrogenase activity detected by the cupric ferrocyanide was localised in the vicinity of the cell membrane and the nucleus. Detection of similar cytochemical reaction products in material subjected to lengthened incubation in the original medium, rather than to transfer (which showed no significant change in the level of spectrophotometrically-detectable NADP-GDH enzyme activity) implies that regulation of the enzyme may include an activation process which is mimicked by the preparation methods employed for electron microscopy. Certainly, care needs to be exercised in interpreting activity measurements of enzymes after fixation treatments (Elhiti, Moore & Butler, 1986).
The copper ferricyanide method was used to localise NAD- and NADP-linked glutamate dehydrogenases during mushroom cap development (Elhiti, Moore & Butler, 1987). The techniques were validated using malate, lactate and succinate dehydrogenases (MDH, LDH, and SDH). Tests were applied to unfixed and formaldehyde-fixed material from three developmental stages of fruit bodies of Coprinopsis cinerea before spore formation (stage 2), with young spores (stage 3), and with mature spores (stage 4) [see Table 2]. Spectrophotometric assay showed that NADP-GDH was still detectable after 15 min formaldehyde fixation and approximately 9 and 80% activity of NAD-GDH and MDH, respectively, was preserved. At all stages of development MDH, LDH and SDH activities were localised (as expected) in mitochondria. In contrast, both NAD-GDH and, especially NADP-GDH, were localised in cytoplasmic vesicles, mainly in basidia and very young spores, sometimes being found adjacent to the cell walls, suggesting that these enzymes are transported to the vicinity of the plasma membrane (Elhiti, Moore & Butler, 1987).
Location close to the membrane and wall would be consistent with enzymological data, indicating that NADP-GDH contributes to an ammonium scavenging system [view now]. However, ultrastructural localisation of this system at the peripheral region of basidia, in particular, gave rise to the idea that this ammonium scavenging system may have more to do with protecting the meiotic apparatus against ammonium inhibition than with the purely metabolic function of assimilating ammonia. In support of this idea we demonstrated that increase in activity of both of NADP-linked glutamate dehydrogenase (NADP-GDH) and glutamine synthetase (GS) was initiated in the fruit body cap as karyogamy became evident in normally-developing basidia of fruit bodies of Coprinopsis cinerea. Derepression of the two enzymes was highly positively correlated (correlation coefficient 0.91), implying co-ordinated regulation (Moore, Horner & Liu, 1987).
In this study we also demonstrated for the first time that application and injection of solutions of ammonium salts to immature Coprinopsis fruit body caps arrested their further development. Other salts had no such effect, indicating a specific sensitivity to ammonium. It was already known that sporulation of the yeast Saccharomyces cerevisiae is also sensitive to inhibition by ammonium salts and similar enzymic events occur during yeast sporulation. We concluded that meiosis generally includes ammonium-sensitive processes and that co-ordinated derepression of the ammonium scavenging NADP-GDH and GS is a device to ensure removal of ammonium ions from the meiotic cell (Moore, Horner & Liu, 1987). Later, we demonstrated that the stage in sporulation between the end of meiosis and emergence and outgrowth of sterigmata from the basidium of Coprinopsis are most sensitive to inhibition by ammonium.
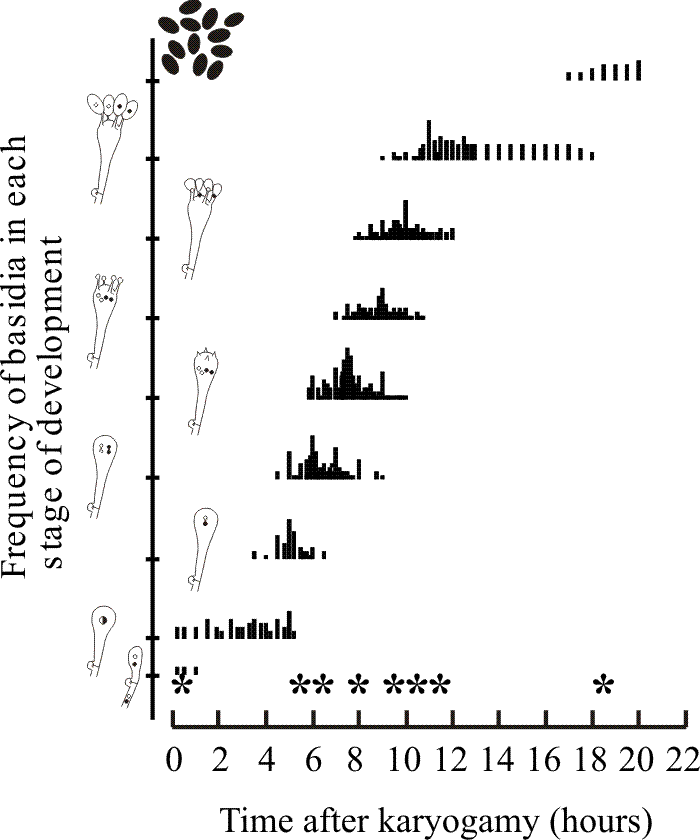 |
| Fig. 20. Time course of progress through meiosis and sporulation in Coprinopsis cinerea. The abscissa shows elapsed time. Slivers of gills were removed from fruit bodies at regular time intervals and sample populations of hymenial cells observed in squashes were quantitatively categorised into the developmental stages represented in the cartoons on the ordinate. The cartoons illustrate the following steps: the basidium arises as the terminal cell of a dikaryotic hyphal branch which inflates and undergoes karyogamy and meiosis. At the conclusion of the meiotic division four outgrowths (sterigmata) emerge from the basidial apex and inflation of each sterigma tip produces the basidiospore (an exospore, produced outside the meiocyte in contrast to the endospores of ascomycetes). Nuclei then migrate from the basidium into the newly formed basidiospores. Mitosis may take place within the basidiospores before they are discharged. Time 0 h is arbitrarily set to the start of karyogamy. Other timings emerged from the observations (as the medians of the distributions shown in the body of the plot): meiosis I occurred at 5 h, meiosis II at 6 h, sterigmata appeared at 7.5 h, basidiospores started to appear at 9 h, began pigmentation at 11 h and mature basidiospores were being discharged from 18 h. The asterisks on the abscissa emphasise these stage medians. Redrawn after Hammad, Ji, Watling & Moore (1993). |
It seems likely that ammonium ions interfere with the cytoskeletal architecture which defines the number, position and nature of the outgrowths which become sterigmata (Moore, Hammad & Ji, 1993). An idea that was further refined when observation of stained nuclei (Fig. 20) coupled with determination of activity of NADP-linked glutamate dehydrogenase (NADP-GDH) in the same tissue, showed that increase in enzyme activity was initiated as karyogamy became evident in normally-developing fruit bodies of Coprinopsis cinerea (Moore, Liu & Kuhad, 1987). Enzyme activity stabilised for about 4 hours during meiosis, but the increase in activity resumed after the second meiotic division, and continued to increase until spore maturation. Parallel assays showed that protein content of fruit body cap tissue declined during development; the decline started before meiosis and also arrested during the division. A major flux in cyclic-AMP content occurred at a much earlier stage, a large accumulation in fruit body initials being rapidly reduced as these developed into primordia. Low levels of cyclic-AMP similar to those recorded in vegetative cells were approached prior to meiosis, suggesting that this nucleotide has little involvement in development of the fruit body after formation of initials. Onset of large-scale utilisation of accumulated glycogen proved to be a post-meiotic event (Moore, Liu & Kuhad, 1987). When the time of exposure to light was varied, karyogamy occurred only in tissues which received at least 24 h light exposure and, most significantly, derepression of NADP-GDH was apparent at the time of or very soon after karyogamy. We concluded that expression of NADP-GDH in the fruit body cap of Coprinopsis is either a component part of the cellular programme involved in karyogamy, or is directly triggered by that programme.
Finally we showed that basidia of Coprinopsis are committed to their developmental pathway, in the sense that they continued through meiosis and sporulation even when excised from their parental fruit body (Chiu & Moore, 1988a). We developed a technique that allowed this differentiation to be demonstrated in vitro. Gill lamellae were removed from caps at various stages of development and placed on the surface of nutrient-agar explantation medium or water-agar. Their development was then observed at daily intervals (Chiu & Moore, 1988a). Continuation of the developmental process after the tissue fragment has been removed from its parent tissue is a classic way of demonstrating embryological commitment. The fact that the basidia of immature gill fragments continued their development, producing mature pigmented spores, is interpreted to mean that once meiosis has started the basidium is committed to complete the sporulation process. These experiments are discussed in detail below [View now].
The experiments show that ammonium ions and glutamine halt meiocyte differentiation and they shed a different light on the function of the NADP-GDH + glutamine synthetase ammonium scavenging system.
Evolutionary Biology of the Fungi The fungi represent superb tools for the study of evolution 'in action'. This 1987 book was the first to bring together, in one volume, coverage of the growing consensus of knowledge and ideas concerning evolutionary biology of the fungi in the widest sense. It draws, not only upon the impetus given to the field by the molecular approaches of the time, but also on the wider technical and philosophical issues raised in the search for evolutionary pattern in fungal life styles, fungal populations and at the phylogenetic level. A Symposium Volume of The British Mycological Society of an international meeting held at the University of Bristol in April 1986, this fascinating text will continue to interest mycologists and evolutionary biologists. |
Rayner, A.D.M., Brasier, C.M. & Moore, D. (eds) (1987). Evolutionary Biology of the Fungi. Cambridge, UK: Cambridge University Press. 465 pp. ISBN-10: 0521330505, ISBN-13: 9780521279253. View Publisher's webpage. |
Chiu & Moore (1988b) discuss reports (see pp. 524/525 in that paper) of similar effects in other organisms, which imply that ammonium inhibition of meiotic processes may be common to a wide range of eukaryotes, though the way (or ways) in which ammonium or glutamine cause inhibition of sporulation processes has not yet been established in any organism. The frequent observation of such inhibitions may suggest action against evolutionarily conserved components of the cell. While there are differences in detail, the common feature is that ammonium and glutamine cause a reproductive state to revert to a purely vegetative one. Inhibitory effects of ammonium and/or glutamine on actin synthesis, wall growth and proton flow have been reported and these phenomena could be rationalised by the suggestion that the inhibitors affect cytoskeletal elements generally concerned with karyogamy and sporulation; the precise expression of the inhibition depending upon the consequential effects of disruption of such cytoskeletal architecture in a given organism.
We have not pursued further this ammonium inhibition of sporulation in Coprinopsis. However, we did investigate a regulatory effect that ammonium exerts on protein utilisation in Ascomycota because of its potential impact on the nutrition of mushrooms. In Aspergillus species, proteinase production is controlled by derepression in the absence of a carbon, nitrogen or sulphur source (discussed, with references, in Kalisz, Wood & Moore, 1987). These organisms secrete at least four proteinases, three of which are different from intracellular proteinases, and derepression involves de novo enzyme synthesis. Proteinase production in Neurospora is controlled by induction and repression. As in Aspergillus, induction involves de novo synthesis, and the secreted proteinases exhibit different properties from intracellular enzymes. The proteinases are induced when protein is present with one of the major elements (carbon, nitrogen, sulphur) otherwise absent. In the presence of protein and all three alternative sources of the major elements proteinase production is repressed in Neurospora. Peptides and amino acids do not cause induction but certain amino acids inhibit proteinase synthesis when added to media deficient in one of the major elements. An ericoid mycorrhizal fungus, Hymenoscyphus (= Rhizoscyphus) ericae (Helotiales, Ascomycota), used protein as a source of both nitrogen and carbon; proteolytic activity being produced in the presence of alternative carbon sources. This aside, a common feature of proteinase regulation in Ascomycota is that proteinase is not produced unless alternative sources of carbon, nitrogen or sulphur are absent (so, for example, ammonium in the medium represses proteinases).
Agaricus bisporus and Volvariella volvacea are grown commercially for food production on composted wheat and rice straw, respectively. Coprinopsis cinerea is a common weed fungus in both cultivations and is a frequent contaminant of mushroom farm beds. Protein is probably the most abundant nitrogen source available to these organisms in compost, in the form of lignoprotein, microbial protein and plant protein. The bulk of compost or straw consists of macromolecules, such as lignin, cellulose, hemicellulose and protein. Lignoprotein polymers constitute the largest compost fraction degraded during mycelial growth of A. bisporus, with cellulose and hemicellulose being degraded during fruiting (discussed, with references, in Kalisz, Moore & Wood, 1986). Compost contains about 10% microbial biomass that probably serves as a concentrated source of nitrogen, carbon and minerals contributing to mushroom growth and yield. A. bisporus is able to degrade dead bacteria and to utilise them as sole source of carbon, nitrogen and phosphorus.
However, Coprinopsis excretes large amounts of ammonium nitrogen into the medium, which implies an ability to utilise the carbon skeletons of amino acids (Stewart & Moore, 1974; Al-Gharawi & Moore, 1977), but might adversely affect utilisation of proteins if proteinase regulation is similar to that observed in Ascomycota. It is important, therefore, that Kalisz, Moore & Wood (1986) demonstrated that all three saprotrophic basidiomycetes (Agaricus bisporus, Volvariella volvacea and Coprinopsis cinerea) are able to use protein readily as a source of carbon, nitrogen and sulphur, and that all three species utilised protein as sole source of carbon, nitrogen and sulphur. Indeed, protein was utilised as efficiently as was glucose when provided as a sole source of carbon. When supplied together as carbon sources both protein and glucose were utilised more rapidly, and growth was greater than when either protein or glucose were supplied separately as sole sources of carbon. Thus no catabolite repression was observed in the presence of glucose and, similarly, no catabolite repression was observed when media were supplemented with ammonium chloride or ammonium sulphate as well as protein. On this basis, therefore, all three mushroom species were judged to be free of the catabolite repression commonly seen in lower fungi. In addition, the production of Coprinopsis proteinase activity was also derepressed in protein-free media in the absence of sulphate; in other words, Coprinopsis proteinase was produced to scavenge for metabolic sulphur if nutrient sulphate was not included in the medium (Kalisz, Wood & Moore, 1987).
Spent culture medium contained five electrophoretically distinct proteolytic activities, which were active at pH 9, when Coprinopsis cinerea was grown in culture medium which contained protein as sole source of nitrogen. The enzymes were separated by ultrafiltration, gel filtration and ion-exchange chromatography (Kalisz, Wood & Moore, 1989). Studies with inhibitors indicated the presence of cysteine-, metallo- and serine-proteinases. Only a few other fungi have so far been reported to produce extracellular cysteine proteinases and the Coprinopsis activity resolved into two proteinases with optimal activities at pH 6 in one case and pH 9 in the other. Although five different proteinases were detected by non-dissociating gel electrophoresis, they all appear to be monomers of about the same molecular weight (31 kDa) which are not glycosylated (Kalisz, Wood & Moore, 1989). Some of the proteinases were active over broad ranges of pH (between pH 5 and pH 11) and temperature (maximal activities at 56° to 62° C). Differences in temperature stabilities and optima of the proteinases may reflect differences in the role(s) of the enzymes in the natural habitats for Coprinopsis. The more thermostable proteinases are perhaps used to degrade proteins in composts, herbivore dung and similar environments at temperatures at which other competitors are unable to thrive. The enzymes, though relatively unstable at higher temperatures, could provide the organism with the required amino acids and peptides before other microbes are able to adapt to these conditions (Kalisz, Wood & Moore, 1989).
As part of our investigations of resource utilisation by commercially cultivated mushrooms, A. bisporus and V. volvacea, and their common weed, Coprinopsis cinerea, we also examined laccase production in various experimental circumstances (Kalisz, Wood & Moore, 1986). In composts, A. bisporus degrades lignoproteins (proteins covalently bound to lignin), and laccases are used to detoxify aromatic metabolites (phenols) formed from lignin degradation. It was already known that lignoprotein polymers constitute the fraction degraded most rapidly during mycelial growth of A. bisporus in compost; with cellulose and hemicellulose degradation dominating during fruiting. These nutritional changes being correlated with changes in extracellular enzyme activities in the compost. During mycelial colonisation a high level of laccase enzyme accumulates, laccase becoming the major excreted protein of Agaricus, this one enzyme activity accounting for 2% of the mycelial protein (0.7% of total fungal biomass). This activity decreases rapidly at the time of fruiting, due to both inactivation and proteolysis. In contrast, cellulase activity remains low during Agaricus mycelial growth, increasing rapidly during fruit body development (see discussion and references in Kalisz, Wood & Moore, 1986). In this study Agaricus bisporus was grown on defined liquid media with protein as sole source of carbon, nitrogen or sulphur and with these nutrients supplied in the form of glucose, ammonium or sulphate. Cultures were incubated at 25°C in the dark in static conditions. Mycelia were harvested at suitable intervals, biomass yields determined and culture filtrates tested for extracellular laccase activity. Constitutive laccase production was observed under all conditions tested. However, Agaricus laccase, though constitutive, was induced by protein and repressed by ammonium. The presence of protein caused a 6-fold increase in the laccase specific activity. Supply of additional ammonium reduced the specific activity by 50%. The A. bisporus laccase, though considered to be constitutive, is clearly strongly regulated in a positive manner by protein and is repressed by ammonium.
Proteinases induced by exogenous substrate proteins may therefore act to stimulate increased laccase secretion as part of an integrated nitrogen recycling mechanism providing for the most efficient utilisation of lignoprotein complexes. Supplementation of these media with ammonium had no effect on the utilisation of protein substrates for growth and had a slightly stimulatory effect on the production of extracellular proteinases by A. bisporus. No detectable extracellular laccase activity was found in similarly tested cultures of Coprinopsis cinerea or Volvariella volvacea (Kalisz, Wood & Moore, 1986). Demonstration of extracellular laccase activity only in the culture filtrates of A. bisporus confirms previous findings that this organism secretes laccase in a constitutive manner and that Coprinopsis spp. and Volvariella volvacea do not produce this enzyme extracellularly. We suggested that this indicates resource specialisation between organisms which otherwise appear to occupy the same habitat (composted plant remains). The idea would be that Agaricus mycelia would be able to attack lignoprotein preferentially, while the weed organism (Coprinopsis) utilises free protein (incidentally, releasing free ammonia into the compost to suppress the competitive abilities of other microbes).
Laccases are also thought to be important contributors to the ability of fungal mycelia to degrade pesticides. Chlorophenols have been frequently used as disinfectants and preservatives but their recalcitrant nature, persistence and toxicities make them priority pollutants for treatment. We tested the ability of various fungi commonly found in wooded areas (Armillaria gallica, A. mellea, Ganoderma lucidum, Lentinula edodes, Phanerochaete chrysosporium, Pleurotus pulmonarius, Polyporus sp., Coprinopsis cinerea and Volvariella volvacea) to degrade pentachlorophenol (PCP) in batch cultures. The PCP content was monitored by reversed phase high performance liquid chromatography, and the breakdown products determined by gas chromatography-mass spectrometry (GC-MS). We also compared the effectiveness of these mycelial batch cultures with the ability of the spent mushroom substrate of the oyster mushroom (Pleurotus pulmonarius), on the grounds that this is a waste product itself which, nevertheless, may have some residual ability to destroy phenolics (Chiu, Ching, Fong & Moore, 1998).
All these fungal mycelia showed active breakdown in addition to biosorption as their PCP removal mechanisms. The tolerance level of the fungus towards PCP did not correlate with its degradative capacity, or to its ligninolytic ability (as determined by ability to decolourise the dye Poly-R478). The A. mellea mycelium showed the highest degradative capacity (13 mg PCP g-1 mycelium dry weight) while the Polyporus strain possessed the greatest biosorption capacity (31 mg PCP g-1 mycelium dry weight). In comparison, the spent oyster mushroom farm compost, which contained a community of both bacteria and fungi, functioned over a wide range of initial PCP concentrations and reached a higher degradative capacity (19 mg PCP g-1 dry weight) in only 3 days. GC-MS chromatograms revealed only residual PCP peaks in extracts of the spent compost, a contrast with the fungal mycelial batch cultures in which a variety of breakdown products were detectable in the culture filtrates. Use of spent oyster mushroom farm compost for bioremediation of biocide-contaminated land-fill sites is a promisingly simple and highly recommendable procedure (Chiu, Ching, Fong & Moore, 1998). [TOP]
Patterns in Fungal Development This account of the developmental biology of fungi considers whether evidence exists for the action of pattern forming mechanisms in the development of fungal structures, similar to those which operate in animal and plant systems. The theory of pattern formation, assumed to be applicable to all multicellular organisms, has been developed largely through the study of animal, and to a lesser extent, plant systems. Fungi, members of the third major kingdom of eukaryotes, have not featured in these studies, although much research of fungal morphology has been undertaken with taxonomic intentions. This first account of the developmental biology of fungal morphogenesis considers whether evidence exists for the action of pattern forming mechanisms in the development of fungal structures. Chapters on the fruit body, on a range of aspects of the hyphae and the mycelium, and on genetic control and nuclear events in morphogenesis provide insights into the mechanisms used in fungal construction. Aimed at developmental biologists as well as mycologists, the terminology has been kept as simple as possible to make the volume accessible to the widest audience. ' … a good accessible book for advanced undergraduates and their seniors, which sets out an area of great opportunity in modern microbiology.' Chris Thurston, in the journal Biologist. |
| Extracts from reviews of: Chiu, S. W. & Moore, D. (1996). Patterns in Fungal Development, Cambridge University Press: Cambridge, UK: Cambridge University Press. 232 pp. ISBN-10: 0521560470, ISBN-13: 9780521560474. [CLICK HERE] to visit the Amazon page. |
[TOP]
Updated December 7, 2016










