13.14 Orchidaceous endomycorrhizas
The Orchidaceae is the largest family of flowering plants, with about 880 genera and 22 000 accepted species, though there may be as many as 25 000). Overall, orchidaceous plants account for around 10% of all seed plants. Just to put the number of orchid species in context, there are about twice as many orchids as there are bird species, and about four times the number of mammal species. Although there are representatives throughout the world; for example, there are about 90 European species, most species are naturally distributed in the tropics and subtropics. However, orchids are commonly cultivated and horticulturists now have more than 100 000 hybrids and cultivars in cultivation around the world.
Orchidaceous mycorrhizas are similar to ericoid mycorrhizas but their carbon nutrition is even more dedicated to supporting the host plant. All orchids have a stage where they are non-photosynthetic and therefore dependent on external sources of nutrients. Orchid seeds are very small and contain little nutrient reserves, so in the vast majority of cases it is the seedling stage that is obligately mycorrhizal. In fact, most orchid seeds will not germinate unless they have been infected by an appropriate fungus, but the young orchid seedling itself is non-photosynthetic and depends on the fungus partner utilising complex carbon sources in the soil, and making carbohydrates available to the young orchid.
All orchids are achlorophyllous in the early seedling stages, but usually chlorophyllous as adults, so in this case the seedling stage orchid can be interpreted as parasitising the fungus. It is now known that many orchid species can be cultured artificially without mycorrhizal infection if they are supplied with exogenous supplies of carbohydrate. Mature orchids with mycorrhizas are more competitive on poor soils.
Another measure of the value of mycorrhizas is that, in vitro, symbiotic seedlings reach a higher nitrogen concentration in their tissues than asymbiotic controls. These observations demonstrate that the mycorrhizal fungi are essential supports for nutrient uptake by the plants.
In the wild, the entry of mycorrhizal fungi into orchid roots was mostly through root hairs. There was no relation between root hair characteristics and the extent of colonisation, and in some epiphytic species fungal entry was directly through the epidermis. The fungi formed highly coiled hyphal structures (pelotons) in the root cortex; peloton size was related to cell size. Fungal invasion of cortical cells was by cell-to-cell penetration. The ratio of intact and lysed pelotons contained in the cortical cells varied between species and life-forms. Chlamydospores and microsclerotial structure were frequently found within the plant’s cortical and root hair cells, being released when the root hairs split open (Sathiyadash et al., 2012).
The mycorrhizal fungi in orchids are Basidiomycota, and in particular, many orchids are associated with species of Rhizoctonia (although this is a complex genus which can be divided into several new genera). Interestingly, many of orchid mycorrhizal fungi are serious pathogens to other plants. This is especially true of Rhizoctonia spp. and Rhizoctonia strains isolated as pathogens of other plants (for example, R. solani, which causes a stunt disease of all the major cereals crops and probably most grasses, and R. cerealis, cause of sharp eyespot of wheat) can support the germination of orchid seeds. Similarly, these and other species of Rhizoctonia can be isolated from mature, and healthy, mycorrhizal orchid roots. However, many of the Rhizoctonia orchid mycorrhizas are saprotrophic, producing a range of carbohydrate-degrading and other enzymes enabling the breakdown of plant debris, including lignin. Some Mycena spp. that associate with Cymbidium and Gastrodia orchids are known better as saprotrophs in leaf litter.
The orchid Gastrodia elata is particularly interesting because its seedlings develop with the saprotroph Mycena osmundicola (a small mushroom, originally found in Bermuda growing on plant litter), but as it matures comes to depend on the highly destructive tree pathogen Armillaria mellea (Honey fungus). Even Lentinula edodes, which is the cultivated shiitake mushroom and is a white-rot wood degrader, can support the development of an achlorophyllous orchid called Erythrorchis, as can several other wood rotting fungi. Thus, orchids use a great diversity of fungi with a range of different nutritional strategies as their mycorrhizal partners (Rasmussen, 2002; Bonnardeaux et al., 2007). The dependency of the plant on its fungi also changes over the lifetime of the orchid; in most orchids decreased dependence is evident from the seedling stage onwards. Some orchids remain greatly dependent on their mycorrhizas in spite of the photosynthetic capacity of their leaves. Generally speaking, seedlings associate with a wider range of fungi than do older plants, suggesting that individual plants change their associations during development.
Infection of an orchid seed by fungi occurs after the embryo takes up water and swells, rupturing the seed coat. The small orchid seeds lack differentiated embryos or food reserves and their germination is limited to an intermediate stage called a protocorm. This may emit a few epidermal hairs before its growth stops. Further development of the protocorm will only occur if a mycorrhizal fungus, like Rhizoctonia, is able to colonise the epidermal hairs (Fig. 5; Fig. 10A-C). An orchid ‘embryo’ consists of only a few hundred cells and the fungi spread quickly from cell to cell. As hyphae penetrate cells of the embryo, the plasma membrane of the orchid cell invaginates, and the hypha becomes surrounded by a thin layer of host cytoplasm.
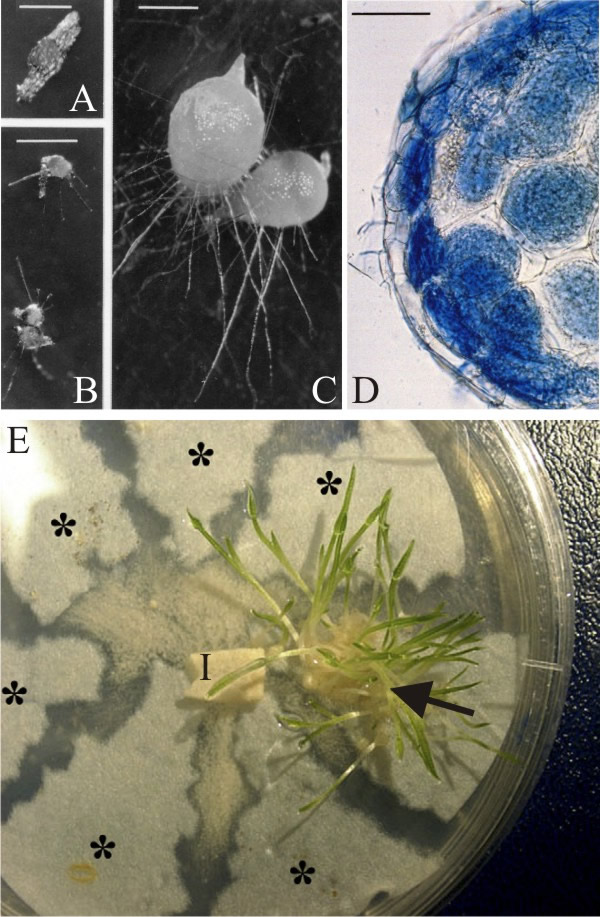 |
|---|
| Fig. 10. Infection of orchid seed by fungi. A-D, Rhizoctonia cerealis forming mycorrhizas with seeds of the heath spotted orchid (Dactylorhiza maculata ssp. ericetorum) on mineral salts agar in the laboratory. A, an ungerminated seed of D. maculata on agar enclosed in its testa (scale bar = 0.5 mm). B, germinated but uninfected protocorms on a control plate. The seeds have increased in size, rupturing their testae, and have produced a few epidermal hairs but no further growth was observed (bar = 1 mm). C, germinated protocorms infected with R. cerealis showing great increase in size and differentiation, polarised and showing evidence of an emerging shoot tip (bar = 1mm). D, section of an infected protocorm mounted in Cotton Blue/lactic acid to stain the fungus blue; note the hyphal coils (called pelotons) in cells of the outer cortex of the protocorm and their disintegration in cells of the inner cortex (bar = 100 µm). E, testing the specificity of orchid-fungus associations by sterile culture assays using orchid seed (on roughly-torn segments of filter paper) and a potential mycorrhizal fungus pregrown on agar medium in a standard Petri dish (original fungal inoculum labelled I). Each piece of filter paper carries a different batch of seed. Only one orchid species is compatible with the fungus grown on this plate and is able to germinate and form seedlings with leaves (arrow). Seeds of other orchids failed to develop (asterisks). A-D modified from Weber & Webster, 2001; E modified from Bonnardeaux et al., 2007; all reproduced with permission of Elsevier. For more information and illustration of mycorrhizas visit Mark Brundrett’s website at http://mycorrhizas.info. |
Within the orchid cells the fungal hyphae form a dense mass of coiled hyphae called a peloton, which greatly increases the interfacial surface area between orchid and fungus (Fig. 10E). Each intracellular peloton lasts only a few days before it degenerates; indeed degeneration may begin within 24 h of formation. The degraded peloton is ultimately left as an amorphous clump of collapsed hyphae surrounded by a continuous sheath of host plasma membrane (called the perifungal membrane). During this process, the plant cell remains perfectly functional and can be recolonised by any surviving hyphae, or by fungi invading from adjacent cells. An orchid cell can go through this cycle several times, being repeatedly re-infected; indeed more than one fungus can form pelotons at the same time in the same orchid tissue.
Traditionally, two types of orchid mycorrhiza have been recognised:
- tolypophagy, which is described above, and is based on degradation of the pelotons; it is found in the great majority of species;
- and ptyophagy, described below, only found in a number of highly mycotrophic tropical orchids.
In ptyophagy the fungal hyphae are digested, older hyphae develop large vacuoles and thickened cell walls, when the cytoplasm degenerates the hyphal cells collapse and are consumed by the orchid cell. In Gastrodia elata, for example, hyphae of Armillaria mellea extend in bundles along the roots in cortical canals that develop from lines of ‘passage cells’ whose adjoining cell walls and original cell content deteriorate. In the host cells outside the passage canal the hyphal coils persist, but the inner cortex contains ‘digestion cells’. When hyphae enter a digestion cell, an interface is formed between the plant plasma membrane and the hyphal wall. The plant releases lytic vesicles into this interface. The fungal wall breaks open in the interfacial space (which is now a digestion vacuole) and invaginations from the plant plasma membrane become pinocytotic vesicles containing fragments of fungal wall which are absorbed by the orchid cell for further digestion (Rasmussen, 2002).
Infection by mycorrhizal fungi does not necessarily result in the germination and growth of an orchid. Upon infection, three outcomes are possible:
- a mycorrhizal interaction, as described above;
- parasitic infection, in which the orchid cells are invaded and the embryo dies;
- the orchid cells reject the fungal infection and fail to establish a mycorrhiza.
A successful fungal infection results in successful development of the orchid seedling. Although the mycorrhizal fungus can be the sole source of nutrition for the orchid during the first years of life, most orchid species develop chlorophyll as they mature and become less dependent on the mycorrhiza. However, most mature plants will still have mycorrhizal roots, using the fungus to enhance nitrogen and phosphorus uptake.
There are about 200 species of orchid that remain achlorophyllous when mature; examples are the genera Galeola, Gastrodia, Corallorhiza, and Rhizanthella. These continue to depend on their mycorrhizal fungi for their nutrition (that is, they are obligate mycoheterotrophs). Some chlorophyllous orchids extend their dependency on their mycorrhizal fungi by spending several years underground before producing aerial (and photosynthetic) flowering stems (for example, the European wild orchids called helleborines); these are partial mycoheterotrophs.
Updated July, 2019
