7.7 Gene segregation during the mitotic division cycle
In the middle of the twentieth century it became evident that meiotic segregations were not the only way of making maps of chromosomes. Mitotic segregations can also be analysed and are a convenient way of mapping chromosomes. The approach is applicable to any fungus that is normally haploid, although the first step is the selection of diploids that arise spontaneously through nuclear fusion at a rate of about one in every 106 or 107 mitoses.
The pioneering work was done with the ordinarily haploid filamentous fungus Aspergillus nidulans. Selection of diploid strains is a little easier in Aspergillus because its conidia are always uninucleate. Consequently, rare diploid conidia can be selected from amongst a large spore population obtained from a heterokaryon by selecting for a heterozygous phenotype. Uninucleate spores cannot be heterokaryotic, so conidia expressing a heterozygous phenotype must contain both homologues of at least one pair of chromosomes, and may be completely diploid. So if you make a heterokaryon between two recessive auxotrophs you would expect that diploid spores would be the only conidia able to grow on minimal medium (see Fig. 2 above). Diploid conidia are larger than haploid conidia, being about twice the volume; they also, of course, contain twice the haploid amount of DNA (see Fig. 1 above) (Stukenbrock & Croll, 2014).
This sort of nutritional selection is an automatic method that certainly works efficiently, but it limits the number of nutritional markers that can be used in any experimental crosses. However, an especially useful feature of A. nidulans (not true for all fungi) is that the colour of the conidium depends on its own genotype. Consequently, a heterokaryon made between two non‑allelic, recessive, spore colour mutants, say white-spore and yellow-spore strains, will produce large numbers of haploid white and yellow conidia together with very occasional sectors of diploid conidia with the wild type dark green colour. Using colour selection leaves open the possibility of having several (unselected) nutritional markers in the cross, but it requires close scrutiny of the cultures.
Nutritional selective methods have been used to isolate diploids from many normally haploid fungi. This includes basidiomycetes such as Schizophyllum commune and the agaric Coprinopsis cinerea, but it is especially important in those fungi in which the known life cycle lacks sexual reproduction, and this group includes several commercially important species, including Aspergillus niger, A. oryzae, A. flavus, Penicillium chrysogenum and plant pathogens like P. expansum and P. digitatum.
Diploids are generally sufficiently stable to grow into diploid vegetative colonies, but these do produce rare sectors showing segregation of the originally heterozygous genes. This type of segregation, also based on mitotic recombination, can be used for genetic mapping. The key to understanding is to remember the crucial differences in chromosome behaviour during meiosis and mitosis. In meiosis, homologous chromosomes undergo synaptic pairing, and they take their place on the first division spindle as a bivalent comprising two chromosomes, each divided into two chromatids (the four-strand stage as illustrated in Fig. 9). At the first division of meiosis, the undivided (that is, maternal and paternal) centromeres are taken to opposite poles of the division spindle. None of this happens in mitosis.
In mitosis, homologous chromosomes do not line up with one another, so there is no synapsis. The fact that recombinant diploid segregants can be obtained from mitotic crossing over indicates that occasional exchanges occur between homologous chromosomes during mitosis, and the recombination results in two homologous chromosomes with reciprocally recombinant arms (Fig. 9), but it is extremely rare. In fact, mitotic crossovers are too rare for double exchanges to be a problem in genetic analysis. Mitotic crossing over can be visualised as very similar to meiotic crossing over, but the consequences in terms of the genotypes of progeny nuclei differs because chromosome segregation differs between meiosis and mitosis. If a crossover takes place in mitosis the two chromosomes involved do not stay together (as they do in meiosis), but they separate and reach the equator of the division spindle independently. The two (homologous) chromosomes, which are, of course, divided into two daughter chromatids, then behave independently.
In meiosis, the two reciprocally recombinant chromatids must end up in different haploid daughter nuclei (Fig. 9). But mitosis produces diploid daughter nuclei by sending one daughter chromatid of each homologous chromosome to each pole of the division spindle. Providing the rule that daughter chromatids must go to opposite poles of the division spindle is upheld, there is no other constraint. The daughter chromatids of a pair of homologous chromosomes segregate independently of each other. Consequently, following a mitotic crossover:
- the two reciprocally recombinant chromatids may pass to opposite poles at the subsequent anaphase stage of mitosis;
- alternatively, and with equal probability; they may go to the same pole.
The importance of this is that in the former case (where a recombinant chromatid is accompanied by a parental chromatid) all markers between the crossover and the end of the chromosome will become homozygous (Fig. 9).
In most organisms, the analysis of meiotic products is usually the easiest way of mapping the genome. However, use of mitotic segregations for genetic analysis does offer some worthwhile advantages over meiotic analysis to the experimenter. Relatively few segregants can provide a considerable amount of information about relative positions of genes on the chromosome. Even one diploid segregant in which linked genes a and b become homozygous simultaneously provides evidence that they are in the same chromosome arm, and a second segregant in which a becomes homozygous alone shows almost certainly that a is distal and b proximal with respect to the centromere.
Meiotic and mitotic linkage maps show the same gene orders but the spacing between the genes differs, implying different distributions of crossovers in mitosis and meiosis. The overwhelming advantage of mitotic analysis, though, is in the formation of haploids. Because mitotic recombination is so rare, genes on the same chromosome show complete linkage during haploidisation. Genes reassort freely if they are not on the same chromosome. Thus, linkage group assignments can be made far more efficiently than is possible in meiotic analysis.
The method was developed first with Aspergillus nidulans, in which segregant sectors of the mycelium could be recognised by the colour of their spores. In A. nidulans, mitotic crossing over has a frequency of about two per thousand mitotic divisions and haploidisation about one per thousand mitotic divisions. In a reversal of the procedure used for identifying diploid sectors, segregants can be identified by the appearance of yellow or white-spored sectors against the background of dark‑green spores of a parental diploid colony heterozygous for recessive colour mutations.
 |
| Fig. 9. . Comparison of the segregation mechanisms of meiosis and mitosis. At the top we show meiosis, on the left without recombination, on the right incorporating a crossover event. These are the perfectly normal meiotic segregation processes applicable to all eukaryotes. Mitosis, with and without a crossover, is shown in the bottom part of the figure. Mitosis starts with replication of the parental chromosomes, but the two homologues do not normally associate with one another. Subsequently, the replicated chromosomes align on the mitotic division spindle independently, and the rule is that one daughter chromatid of each replicated chromosome segregates into each daughter nucleus, so that the two daughter diploid nuclei have the same genotype as the parent. Very rarely, the replicated chromosomes do associate with one another sufficiently closely for a crossover to occur. This results in two of the daughter chromatids being recombinant. However, the recombinant replicated chromosomes still independently align on the mitotic division spindle, and still follow the rule that one daughter chromatid of each replicated chromosome segregates into each daughter nucleus. Because of the crossover, there are two possible orientations on the mitotic division spindle. One produces two diploid daughter nuclei that are homozygous from the point of the crossover to the end of the chromosome arm (one homozygous mutant, one homozygous wild type). The alternative orientation produces two heterozygous diploid daughter nuclei, but one contains the two parental chromatids, and is exactly the same as the parental nucleus, whereas the other receives the two recombinant chromatids. Modified from Chapter 5 in Moore & Novak Frazer, 2002. |
We will examine some results from a typical experiment with Aspergillus nidulans in which the original diploid was heterozygous for both white (w) and yellow (y) conidia. These two genes are on different chromosomes, and the y chromosome also carried auxotrophic mutations (that is, with a nutritional requirement because of their mutation) called ade (adenine requirement), pro (proline requirement), paba (requirement for the vitamin para-aminobenzoic acid), and bio (requirement for the vitamin biotin). The parental diploid had the chromosomal constitution shown in Fig. 10: phenotypically, it had wild type green (diploid) spores and could grow (as a diploid mycelium) on minimal medium with no nutritional supplements.
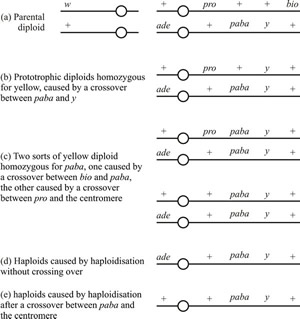 |
Fig. 10. Yellow-spored mitotic segregants obtained from an experiment with Aspergillus nidulans in which the original diploid was heterozygous for both white (w) and yellow (y) conidia. The parental diploid had the chromosomal constitution shown in Fig. 10a, and the crossovers referred to in the other sections of the figure occurred in one chromosome of this genotype before mitotic segregation. The open circle represents the centromeres; + represents the wild type alleles; ade = mutant with a nutritional requirement for the purine adenine; pro = nutritional requirement for the amino acid proline; paba = nutritional requirement for the vitamin para-aminobenzoic acid; bio = nutritional requirement for the vitamin biotin. Modified from Chapter 5 in Moore & Novak Frazer, 2002. |
Segregants from this diploid were identified on the basis of spore colour: the parental diploid produces dark green conidia, but sectors with yellow spores and sectors with white spores are occasionally found. Amongst the yellow-spored segregants (Fig. 10) were strains which were:
- prototrophic diploids homozygous for yellow, caused by a crossover between paba and y (Fig. 10b);
- two sorts of yellow diploid homozygous for paba, and therefore auxotrophic for para-aminobenzoic acid, one caused by a crossover between bio and paba in the parental diploid, while the other was caused by a crossover between pro and the centromere (Fig. 10c);
- haploids caused by haploidisation without crossing over (Fig. 10d);
- haploids caused by haploidisation after a crossover; in this case a crossover somewhere between paba and the centromere (Fig. 10e).
White-spored segregants resulted from homozygosity or haploidisation of w (Fig. 11). White-spored genotypes observed were:
- prototrophic white-spored diploids caused by a crossover between w and the centromere (Fig 11b);
- two sorts of white-spored haploids caused by haploidisation without crossing over (Fig 11c).
White-spored haploid segregants requiring proline and biotin were observed in about the same frequency as those requiring adenine and p-aminobenzoic acid, showing that the chromosome carrying these auxotrophic markers segregated independently of the white chromosome during the haploidisation process.
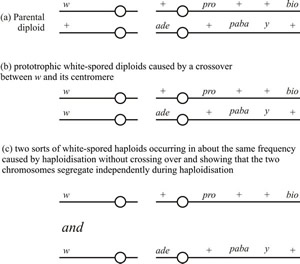 |
Fig. 11. White-spored mitotic segregants obtained from an experiment with Aspergillus nidulans in which the original diploid was heterozygous for both white (w) and yellow (y) conidia. The parental diploid had the chromosomal constitution shown in Fig. 11a, and the crossovers referred to in the other sections of the figure occurred in one chromosome of this genotype before mitotic segregation. Modified from Chapter 5 in Moore & Novak Frazer, 2002. |
These segregants, the yellow diploid segregants in particular, show that mitotic crossing over is a reality, so let’s see what lessons can be learned from this example.
For practical analysis, the rule is that in diploid segregants homozygosis for one gene will always be accompanied by homozygosity of any genes distal to it (that is, on the side closest to the chromosome end) in the same chromosome arm, but not necessarily by homozygosity of genes more proximal to it (that is, on the side towards the centromere). This general characteristic of mitotic segregation is what allows us to deduce the genetic map of the segment of chromosome involved, and is illustrated in the yellow diploid segregants in Fig. 10. Homozygosity for y was invariably accompanied by homozygosity for the wild type allele of bio. This fact places the bio gene distal (that is closer to the end of the chromosome) to y in the same arm of the chromosome. Similarly, homozygosity for paba was frequent but not invariable in diploids homozygous for y, while homozygosity of pro was even less frequent in the yellow homozygotes. This pattern indicates that paba and pro were proximal to y in the same arm; that is, on the centromere side of y, with pro the nearer to the centromere. Noting that co-segregation of ade and paba in yellow haploids suggests that the two genes are linked, the absence of yellow diploid segregants homozygous for ade allows us to infer that this gene may be in the other arm of the chromosome (other data confirm that homozygosis of one arm of a chromosome occurs independently of homozygosity of the other arm).
The frequencies of the different sorts of homozygous diploid segregants are a measure of relative map distance between the genes; but it must be stressed that mitotic recombination is rare, so it is not easy to assemble a sample of independent segregants large enough to make frequency measurements reliable. Nevertheless, even a few segregants can give absolute guidance about gene order relative to the end of the chromosome arm.
Updated July, 2019
