7.1 Compatibility and the individualistic mycelium
Because of the difference in ploidy, the life cycles of true fungi and those of other major groups of eukaryotes differ significantly. For example, for most true fungi diploid nuclei are only produced transiently during sexual reproduction, whereas the haploid state is limited to the gametes in most animals and plants. For true fungi there is great variation in how the process of sexual reproduction is achieved, that is in the mechanics of the process; and there is also a great variation in the duration of each phase of sexual reproduction. Indeed diploids even occur in nature. A study that collected 154 isolates of Aspergillus nidulans from nature found that 140 were haploid strains and 14 were diploid. The diploid strains were not stable in cultivation, though, and eventually became haploid.
When they are formed, whether naturally or by some experimental manipulation, diploid nuclei are larger than haploids and the nuclear-cytoplasmic ratio is correspondingly higher (Fig. 1).
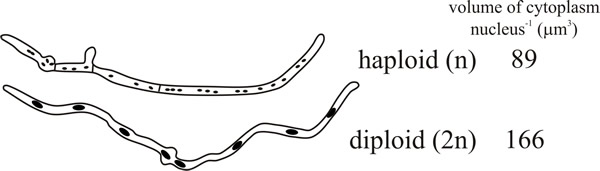 |
| Fig. 1. Diagrammatic comparison of germinating haploid and diploid spores of Aspergillus nidulans. Redrawn from camera lucida drawings in Fiddy & Trinci, 1976. |
One consequence of being haploid is that all mutations can be expressed as there is only one copy of each chromosome in the nucleus. However, as there are many nuclei in a common cytoplasm, mutations occurring in individual nuclei may not be expressed because any lost functions will be provided by the many normal nuclei in the same hyphal system.
The fungal kingdom is very large and diverse, and as a consequence of this the part played by sexual reproduction in the life cycle of fungi is also very diverse. At one extreme of the spectrum of behaviour, fungi display many forms of sexuality that govern the bringing together of genetic information from different parents into some arrangement that eventually produces a (potentially heterozygous) diploid nucleus. This nucleus undergoes meiotic division during which chromosomal segregation and genetic recombination take place as in every other eukaryote. At the other extreme, there are fungi that are completely asexual organisms. In the past, most examples of these have been separately classified and informally named deuteromycetes, imperfect fungi, and anamorphic fungi, or were given more formal taxonomic names such as Fungi Imperfecti or Deuteromycota, but molecular methods have allowed them now to be placed alongside their sexually-reproducing relatives, and such informal assignments are no longer warranted (Seifert & Gams, 2001).
However, some of the most frequently encountered fungi are asexual, and many of them are of enormous economic significance, either as industrial fungi or as pathogens. As fungal taxonomy is still based on the morphology of the sexual reproductive structures, it is best to use informal descriptions such as deuteromycetes, imperfect fungi, anamorphic fungi (which is used generally to indicate the asexual or imperfect state), or mitosporic fungi (which refers to their spores being formed by mitosis). Anamorphic fungi can be yeasts or filamentous forms, and the latter fall into three morphological groups known most commonly as:
- hyphomycetes, which are mycelial forms producing conidia on separate hyphae or groups of hyphae but not in fruit bodies (Bärlocher, 2007);
- coelomycetes form conidia in fruiting bodies that may be called pycnidia, acervulae or stromata;
- agonomycetes, which do not form conidia but may produce chlamydospores, sclerotia or other vegetative structures.
As knowledge accumulates, an anamorphic fungus may be classified into the Ascomycota or Basidiomycota following phylogenetic analyses or discovery of a sexual phase (known as the teleomorph). For example, a particularly important anamorphic fungus, the human pathogenic yeast Candida albicans, has been shown by genomic comparisons to possess homologues of genes known to be involved in meiosis in Saccharomyces cerevisiae and some other eukaryotes, which implies that C. albicans may have a complete sexual cycle.
Many fungi long thought to be anamorphic have been found eventually to have a sexual stage. Candida glabrata remains as a potentially asexual fungus. Previously known as Torulopsis glabrata, Candida glabrata is a highly opportunistic pathogen of the human urogenital tract and the bloodstream and is especially prevalent in elderly and immunocompromised patients. Despite major efforts, no sexual cycle has yet been found, but all known isolates are haploids of one or the other of two opposite mating types and much of the machinery associated with mating and meiosis are present in the genome; indeed, there is evidence of genetic recombination in the population. It is believed that anamorphs have arisen by the loss of sexuality, which has occurred in many different groups of fungi at different time as an adaptation to the lifestyle represented by asexual vegetative growth. It seems that mutational loss of sexuality results in a competitive advantage for the asexual derivatives; because they no longer need to express the numerous mRNAs and proteins required for sexual reproduction (Heitman, 2015).
Fungal genomes vary considerably in size and organisation; they have an average genome size of about 37 Mb, but size ranges from 2.3 Mb for the microsporidian Encephalitozoon intestinalis, while the genomes of several basidiomycetes and ascomycetes greatly exceed 100 Mb, with the largest genome on record at the time of writing being the approximate 8000 Mb for Entomophaga aulicae, (Gregory et al., 2007; and see Section 5.7). Similarly, chromosome numbers and ploidy levels can differ even between closely related species. Differences in the genomes between fungi are a reflection of the interaction of mutational processes with the life style and population biology of the different species. Even the asexual organisms to which we have just referred are not static in an evolutionary sense because they can generate variation by modifying genetic expression or by adapting processes that occur during the mitotic division to produce asexual propagules in which chromosomes have segregated in new combinations or which contain recombinant chromosomes (Taylor et al., 1999; Pringle & Taylor, 2002; Stukenbrock & Croll, 2014) (see Section 7.7 below; CLICK HERE to see it now).
Before we can take the discussion very much further, we have to define some of our vocabulary. A homokaryon is a mycelium in which all nuclei are of the same genotype; in a heterokaryon, the nuclei in the mycelium have two or more genotypes. A species is homothallic if an individual can complete the sexual cycle on its own, but as we will explain below, there are different ways in which homothallism can be achieved. We should also emphasise that a homothallic species is not limited to self-fertilisation. Two homothallic strains may well interbreed, either in nature or with ‘assistance’ in the laboratory. The point is that a homothallic has the ability to self-fertilise, it is not a necessity.
In contrast, for a heterothallic species, sexual reproduction requires interaction of two different individuals. Individual isolates of heterothallic fungi are self-sterile or self-incompatible, but can be cross compatible. Heterokaryosis results from the fusion of hyphae of different isolates, followed by migration of nuclei from one hypha into the other, so that the hyphae come to have two kinds of nuclei, and such a hypha is a heterokaryon.
In the most highly adapted version of this behaviour, in model basidiomycetes like Coprinopsis cinerea and Schizophyllum commune, a basidiospore germinates to produce homokaryotic mycelium with uninucleate cells, called a monokaryon. When two monokaryons meet, hyphal anastomoses occur, and, if they are vegetatively compatible, nuclei of one migrate into the mycelium of the other, the dolipore septa between the cells being broken down to allow nuclei to migrate. If, in addition, the nuclei have compatible mating types, the new growth and the cells of much of the pre-existing mycelium will form binucleate cells, containing one nucleus of each monokaryotic parent; this mycelium is called a dikaryon.
However, there are some important differences in life style between higher fungi and other eukaryotes that have great genetic significance. First, many fungal mycelia can tolerate (in fact, more than tolerate, can benefit from) the presence of several genetically distinct nuclei within their hyphae. This probably arises from the second important difference that we have already mentioned, namely the fact that hyphal anastomoses occur very readily within the Ascomycota and Basidiomycota but are not observed in the zygomycetes.
An important benefit of heterokaryosis is that recessive mutations will not be expressed in a heterokaryon if sufficient nuclei containing the dominant alleles are present (and unless manipulated by an experimenter, recessive mutations are always in the minority). The presence of unexpressed recessive mutations in a population of nuclei in a heterokaryon means that such mycelia have a larger gene pool than homokaryons and this provides the potential for evolutionary selection and hence a more immediate response to environmental stress. Thus, despite being haploid, true fungi are a successful group and this success can be attributed in large part to their use of heterokaryosis to overcome the genetic and physiological disadvantages associated with having just a single nuclear genotype (whether haploid or diploid) within the vegetative growing body. The possibility has also been pointed out that the DNA of supernumerary nuclei in filamentous fungi could serve as a store of nitrogen, phosphorus and carbohydrate, which could be degraded by autophagy with the breakdown products recycled, and providing nutrition for hyphal tips to continue foraging into new areas (Maheshwari, 2005); we’re not sure how likely this is, but supernumerary nuclei must have some selective advantage.
Updated July, 2019
