7.4 The dikaryon
In the Basidiomycota the heterokaryon is highly specialised, being made up of cells that each contains nuclei from two compatible mating types within a common cytoplasm; this is called a dikaryon. A similar arrangement exists in the ascogenous hyphae of Ascomycota, but in many Basidiomycota the dikaryotic condition is purposefully perpetuated by a special kind of hyphal growth (Fig. 6) so that the dikaryotic mycelium can grow indeterminately like any other mycelium. Under normal circumstances only dikaryotic mycelium forms fruit bodies.
The dikaryotic state is maintained by a specialised cell biology: the nuclei divide together (= conjugate mitosis) and at the same time a ‘clamp connection’ grows out as a backward projecting branch which loops backwards and then fuses with the parent hypha. One of the nuclei completes its mitosis in the clamp connection, the other nucleus stays in the main body of the hypha. This separation means that two normal dolipore septa can be formed between daughters of the two mitotically-dividing nuclei and both terminal and subterminal cells (that is, the two daughter compartments) each contain two nuclei of opposite mating type; one nucleus of the subterminal cell being delivered to it through the clamp connection (Fig. 6B). The regularity of this process results in every cell of the dikaryon containing two sets of homologous chromosomes: that is, the dikaryon is a functional diploid.
This is an important point. The so-called ‘higher organisms’ (plants and animals) are diploid but most true fungi are haploid, and this has genetic and evolutionary consequences. However the haploid Basidiomycota have developed functionally diploid cells by extending the vegetative life of dikaryotic heterokaryons and in so doing they have gained the genetic (and evolutionary) advantages of diploid organisms, whilst remaining haploid. Like the diploid, a dikaryotic cell also has two sets of homologous chromosomes, even though each set is located in a separate nucleus. Together, the two nuclei control and regulate the activities of a specific volume of cytoplasm. Complex cellular events have been evolved by Basidiomycota to establish and maintain the dikaryotic condition. These include different mating types and vegetative compatibility systems, elaborate dolipore septa, and an elaborate cytokinesis mechanism in which a backwardly directed hyphal branch enables a daughter nucleus to leapfrog a septum and maintain the dikaryotic condition in each daughter cell of the mitosis (Fig. 6). No wonder Basidiomycota are considered the most advanced group of fungi.
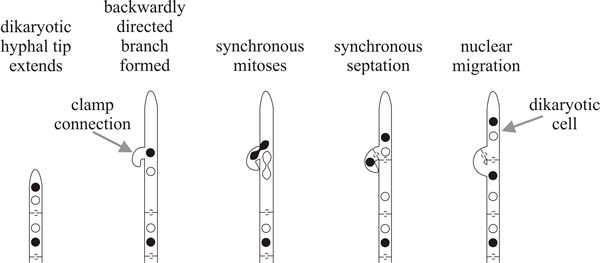 |
| Fig. 6. The dikaryon: a specialised binucleate heterokaryon. The dikaryotic state is maintained by the clamp connection: a backward-projecting hyphal branch which fuses with its parent hypha to deliver one of the nuclei and ensure that each compartment contains two nuclei of opposite mating type separated by a dolipore septum. Each hyphal compartment therefore contains two sets of homologous chromosomes and is a functional diploid. |
Clamp connections are required to maintain the dikaryon because the dolipore septum controls organelle migration and normally hinders proper nuclear distribution. Clamp connections may not be necessary if the hypha is so wide that the conjugate mitoses can take place without hindrance, so some vegetative dikaryons may not form clamp connections. For most of the Basidiomycota, only the dikaryotic mycelium can differentiate into a fruit body, but many fruit bodies contain specialised tissues in which the dikaryotic condition breaks down. For example, many cells in the stem of the Ink cap mushroom fruit body of Coprinopsis are enormously enlarged and are multinucleate, whereas cells elsewhere in the same fruit are regularly dikaryotic. Similarly, not all basidiomycete mycelia are dikaryotic; some are multinucleate, and it is curious that more fungi have not become true diploids. Perhaps the more flexible heterokaryotic condition offers greater advantages than we suspect.
Dikaryon formation occurs via the activities of cell type-specific homeodomain transcription factors (the mating type factors), which form regulatory complexes to establish the dikaryotic state. Many years of classical genetic studies with Coprinopsis and Schizophyllum mushrooms created the foundation of our understanding of how the mating type factors work. Basidiomycota includes many pathogenic fungi too, including the corn (smut) pathogen Ustilago maydis and the globally distributed human pathogenic Cryptococcus neoformans, a leading cause of fungal meningitis. These two have featured in more recent molecular studies that have revealed novel mechanisms of regulation that function downstream of classic homeodomain complexes to ensure that dikaryons are established and propagated (Kruzel & Hull, 2010). For example, in Ustilago maydis, dikaryon formation is controlled by a DNA damage response cascade that includes two conserved DNA-damage checkpoint kinases, called Chk1 and Atr1 (Pérez-Martín & de Sena-Tomás, 2011).
Updated July, 2019
