15.2 Ant agriculture
Attine ants are a group of more than 200 fungus-growing ant species living in the Neotropics (Central and South America) that have a rather unusual ability to cultivate fungi. Most use leaf-litter debris for fungal cultivation, but the leaf-cutter ants (Atta spp. and Acromyrmex spp.) cut and collect fresh leaves to grow two genera of fungi, Leucocoprinus and Leucoagaricus in the family Agaricaceae (Basidiomycota: Agaricales) (Mueller & Rabeling, 2008). This ant-fungus mutualism shows how successful, and how complicated, a relationship like this can be (Mueller et al., 2001; Sánchez-Peña, 2005).
Fungus cultivation evolved apparently only once in the attines, about 45-65 million years ago. The ants actively inoculate their nest with the fungus and then cultivate it by providing it with pieces of leaves, pruning the hyphae and removing intruder fungi. As a reward, the fungus provides bundles of specialised hyphae that the ants use as a food source; these are the gardening ants. The ants are engaged in an agricultural activity; they collect fresh leaf biomass to convert it to compost in order to cultivate a particular fungus that then provides the main food source for the nest (Vega & Blackwell, 2005). Schultz & Brady (2008) point out that agriculture is a specialised symbiosis that is known to have evolved in only four animal groups: ants, termites, bark beetles, and humans.
Gardening ants, which collect and compost plant material, occur in Central and South American tropical rain forests where they are the dominant herbivore (only humans destroy more trees). They destroy the forests, of course, but also damage crops, devastating tracks up to 30 cm wide. An ant colony may contain several million ants, cover 8 m2 and may be 1 m deep. Leaf cuttings (several mm square) are pulped by ants and the chewed-up plant material + saliva + faeces are turned into compost for a fungal garden which is spawned with mycelium carried from parts of an established garden. The garden is a monoculture of a member of the order Agaricales which does not normally produce fruit-bodies. Under the care of the ants, the mycelium produces hyphae with swollen tips rich in lipids and carbohydrates (bromatia; these are also called gongylidia, both words mean ‘swollen hyphal tips’ although, strictly speaking, bromatia are produced in ant gardens and gongylidia in termite gardens) (Fig. 1).
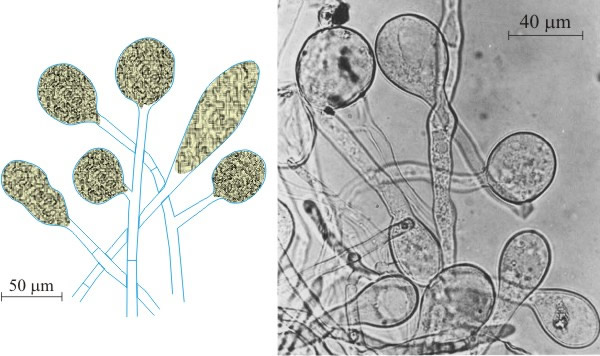 |
| Fig. 1. Diagram and micrograph of bromatia (inflated hyphal tips) of the ant-cultivated fungus, Leucoagaricus gongylophorus (Agaricales), which are the swollen tips of hyphae. They provide the main food of the ants because they contain accumulations of carbohydrates, particularly trehalose and glycogen (photograph by Jack Fisher, from Fisher, Stradling & Pegler, 1994; reproduced with permission of Elsevier). These are also called gongylidia, both words mean ‘swollen hyphal tips’ although, strictly speaking, bromatia are produced in ant gardens and gongylidia in termite gardens. |
These swollen hyphal tips are cropped by ants and are the main food for the larvae; the larvae depend entirely on the fungus to digest the leaves (Bass & Cherrett, 1996). The first genomic studies of the attine leaf-cutting ant genera Atta and Acromyrmex demonstrated losses of two genes in the arginine synthesis pathway (argininosuccinate synthase and argininosuccinate lyase) although all other (non-leaf cutting) ants, including species in the same subfamily (Myrmicinae) as the attine ants, have functional copies of those genes.
When transcriptomes of four different attine ant species (three species of the genus Sericomyrmex and Apterostigma megacephala) were specifically searched for genes coding for argininosuccinate synthase and argininosuccinate lyase, they were not found. So, there is strong genomic and transcriptomic evidence that loss of the ability to produce the amino acid arginine is the reason for the obligate dependence of attine ants on their cultivated fungi; The evidence also strongly suggests that the loss coincided with the origin of attine ants (Nygaard et al., 2011; Suen et al., 2011; Ješovnik et al., 2016).
The fungus must be carried from nest to nest and this is done by newly-mated females as part of the mating flight. The nest is started off by a single queen with fungus in the infrabuccal pocket (a filtering device located in the oral cavity of all ants). The new queen mixes the fungus inoculum she carries with some suitable plant material, and lays eggs on it as soon as the fungus begins to grow. She then lays about fifty eggs each day. The first to hatch become workers who eventually establish a nest with a thousand or so interconnected chambers which might be excavated deep into the forest soil and be able to house a colony of several million individuals. When the first workers emerge, foraging and collection of leaves begins. Foraging may be in nocturnal or diurnal cycles depending on species, habitat and environmental conditions.
The workers cut pieces from leaves on trees and carry them back to the nest (Fig. 2A). Because they usually carry the leaves in their mandibles so that the leaf extends over the ant’s head (Fig 2B), they are also called parasol ants. The caste which produces the largest animals among the several million ants in an average nest is the soldier; a 20 mm long ant which is responsible for protecting the colony and its trails against intruders. The most numerous caste in the colony is the worker caste (these ants are about 8 mm long) which forage in the forest in search of leaves. They can cut leaf pieces bigger than themselves and then carry them back to the colony. Leaves are collected along trails within a colony’s territory. Trails make it easier to find resources and reduce aggression between neighbouring colonies. Physical trails are lines of cleared ground that increase efficiency of foraging. There are also chemical trails of pheromones that the ants leave to mark the routes to good foraging. Once delivered to the nest, smaller workers, about half the size of the foragers, chew the leaves into smaller pieces and carry it into brood chambers.
 |
| Fig. 2. . Leaf-cutter ants. Workers (which are about 8 mm long) cut leaves (A), then transport the leaf fragments to their nest (B) (the large size of the leaf fragments they carry is the origin of the alternative common descriptive name ‘parasol ants’). In the nest the leaves are used as compost to grow fungus for food as described in the text (shown as the mycelial background in C). Leaf-cutter ant queens (C) are among the most fertile and long-lived queens of all social insects. Photographs by Alex Wild (http://www.alexanderwild.com/). Figure and legend adapted from Mueller & Rabeling, 2008; © (2007) National Academy of Sciences, USA. |
Then the smallest ants, only 1.5 mm long, take over. These are the cultivators of the fungus garden; the mushroom farmers. They clean the leaf pieces and then inoculate them with fungus mycelium taken from the existing garden. The ants pluck hyphae from existing mycelia and transfer them to new areas of chewed-up leaf substrate in order to expand the garden. The cultivators continuously maintain the fungus garden, but they also tend the larvae and the queen (Fig. 2C). Substrate is usually exhausted and dumped after three or four months. Exhausted substrate is deposited into dump chambers in the nest together with other refuse such as dead ants. Refuse can also be taken from garden chambers and emptied outside the nest.
Resources Box Leaf Cutter Ants video If you have 8 minutes to spare, you might like to view this WMV video showing leaf cutter ants in action |
The fungus cultivated by leaf-cutter ants does not produce spores but it does have those special hyphal tips (Fig. 1) that exude a sort of honey-dew which the cultivator ants collect and feed to the larvae. The fungus concerned in these associations is always an agaric mushroom fungus but not all of them can be found living free in the forest; specifically, the leaf cutter ant fungus is always associated with leaf cutter ant nests. This is a mutual absolute dependence.
The demand for leaf material as the colony grows is enormous. In the tropical rain forests of Central and South America, leaf-cutter ants are the dominant herbivores. That ‘dominant’ label includes the humans of the forest. Around fifty agricultural and horticultural crops and about half that number of pasture plants is attacked. None of this is new, of course. In the last quarter of the nineteenth century leaf cutting ants were described as: ‘... one of the greatest scourges of tropical America...’ and early Brazilian farmers were so frustrated in their battles against leafcutter ants that they concluded ‘Brazil must kill the ants or the ants will kill Brazil’ (Mueller & Rabeling, 2008).
It has been calculated that leaf-cutting ants harvest 17% of total leaf production of the tropical rain forest. Nests located in pastures can reduce the number of head of cattle the pasture can carry by 10 to 30%. Statistics like this reveal how leaf cutting ants can become dominant exploiters of living vegetation and how they impact human agriculture. Leaf-cutting ants compete successfully with humans for plant material and are, therefore, counted as important pests. Because of their foraging activities, leaf-cutting ants can cause production losses in cacao and citrus fruit of 20 to 30%. Losses caused by leaf-cutting ants (assuming no control measures are used) could exceed $US 1 billion per year and this justifies their being described as a ‘dominant herbivore’ (Boulogne et al., 2014).
The combination of a top-of-the-range social insect with a top-of-the-range fungal plant-litter degrader seems to be the key to this success. The social insect has the organisational ability to collect food material from a wide radius around its nest; but the extremely versatile biodegradation capability of the fungus enables the insect to collect just about anything that’s available.
The total number of species of trees per hectare in most plant communities increases from the poles to the equator. For example, coniferous forest in Northern Canada will have 1 to 5 species ha-1, deciduous forest in North America, 10 to 30, but tropical rain forest in South America has 40 to 100 tree species ha-1.
The tropical rain forest has enormous chemical and physical diversity in its plants and this presents a major problem to herbivores. Most plant eaters have a narrow diet tolerance because evolution has equipped them with only a limited range of digestive enzymes. Plant-eating insects usually only eat one plant. The leaf-cutting ants of the tropical rain forest have, on the other hand, a very wide breadth of diet. These ant colonies are able to harvest 50 to 80% of the plant species around their nests. This is almost entirely due to the broad range degradative abilities of the fungus they cultivate.
This is an obligate symbiotic association between ant and fungus. Ant faeces contain nitrogen sources (allantoin, allantoic acid, ammonia and over 20 amino acids), which supplement the compost and are used by the fungus. A proteolytic enzyme produced by the fungus is acquired, accumulated and transported by ants and deposited in faeces; it hydrolyses leaf proteins.
Cellulases produced by the fungus digest cellulose and the products are converted to the fungal carbohydrates that the ants harvest (like other animals, the ants can digest glycogen, but they cannot digest cellulose). So the fungus enables the ants to use plant materials as nutrients, but the ants provide the fungus with a far wider range of plant materials than any plant pathogenic fungus can attack. Some plants do protect themselves by producing deterrents which inhibit cutting, pick-up or feeding. These protectants include toughness, production of sticky latex, and a wide range of defensive chemicals.
There’s a certain irony in the fact that the tropical rain forest is lush and green because all those mycorrhizal fungi in the roots of the trees give the plants that extra something that enables them to grow with tropical exuberance (see Chapter 13). And then along comes a six-legged army of harvesters to cut down all those lush green leaves. To do what? To feed another fungus, that’s what!
Schultz & Brady (2008) analysed molecular phylogenies covering the entire fungus-growing ant tribe (Attini); their data are fossil-calibrated (so they have a reliable time-scale) and involve multiple-genes (so they are a reliable sample of the genomes). They showed that ant agriculture originated with the cultivation of several species of fungi over 50 million years ago in the early Eocene, the second epoch of the Paleogene Period in the Cenozoic Era (Schultz & Brady, 2008) (see Fig. 2.6 for a geological timescale). Ješovnik et al. (2016) reported slightly older dates than these, so the best estimate is that about 65-55 million years ago ant agriculture, as practised by the fungus-farming ‘attine’ ants, arose in the wet rainforests of South America. In contrast, the transition from lower to higher agriculture (see bullet points below) is most likely to have occurred in a seasonally dry habitat, which would have been inhospitable to the independent free-living growth of the fungus cultivated by attine ants. In other words, dry habitats provided selection pressure favouring fungal cultivars specialised for growth in the ant gardens (Branstetter et al., 2017).
Most of the fungi involved in ant-fungus associations (generally described as ‘leucocoprineaceous fungi’) belong to the tribe Leucocoprineae, which forms a large monophyletic clade in the Agaricales (Basidiomycota) made up mainly of the genera Leucoagaricus and Leucocoprinus (Mehdiabadi et al., 2012). Approximately 60 million years ago the Attine ants split into Paleoattine and Neoattine sister clades and during the past 30 million years, three major ant agricultural systems have diverged from the original Paleoattine system, each involving distinct leucocoprineaceous fungal cultivars. Schultz & Brady (2008) categorised ant agriculture into five different types as follows:
- Lower agriculture, practiced by 76 species of ants in the majority of attine genera, including the most primitive, which cultivate a wide range of fungal species; these are the Paleoattine ants, and all the fungi involved in this category are still able to grow in nature independently of ant garden cultivation.
- Coral fungus agriculture, which evolved about 15 million years ago, is employed by a few ants in the ant genus Apterostigma that cultivate a clade of coral fungi (family Pterulaceae; which is a small, mainly tropical, family of wood and leaf-decomposing fungi) closely related to the fungal genera Pterula and Deflexula (Munkacsi et al., 2004).
- Yeast agriculture evolved about 20 million years ago and is used by a few species in the ant genus Cyphomyrmex that cultivate a distinct clade of dimorphic leucocoprineaceous fungi that grow wild as mycelia but as yeast morphs when associated with ants, the gardens being clusters of small, irregularly shaped nodules about 0.5 mm diameter composed of single-celled yeasts (even though the yeast mode of growth is unusual in the Agaricales).
- Generalised higher agriculture, utilised by species in the genera of non-leaf-cutting ‘higher attine’ ants (Sericomyrmex and Trachymyrmex), which cultivate another distinct clade of leucocoprineaceous fungi so adapted to garden life that they are not found outside the ant nest (though they can fruit from nests reared in the laboratory).
- Leaf-cutter agriculture, evolved from within the higher agriculture ants between 8 and 10 million years ago, and is practiced by species of ecologically dominant ants in the genera Atta and Acromyrmex that cultivate a single highly derived species of fungus, which has been identified as Leucoagaricus gongylophorus (Pagnocca et al., 2001).
An interesting feature of molecular studies is that the phylogenetic relationships of the attine ants are mirrored in the phylogenetic patterns among their fungal cultivars (they are said to be congruent) and the congruence might even extend to a fungal parasite of the gardens; Currie et al. (2003) illustrated the relationship as shown in Fig. 3. The evolution of mutualist partners can take two extremes (or something between):
- Co-evolution may be tightly integrated, that is, an evolutionary change in one partner results in a simultaneous evolutionary change in the other.
- At the other extreme, frequent switching between partners can weaken the associations between any given pair of partners, causing a diffuse association between species.
Extensive phylogenetic data indicate that these ant-fungus partnerships have been stable over evolutionary time spans of several millions of years. This historical pattern of long-term ant-fungus fidelity for several million years provides the opportunity for coevolution, in which evolutionary modification in an ant host can prompt modification in its fungal symbiont, and vice versa. Overall it seems that attine ants and their fungal partners represent the full spectrum from ‘one-to-one’ coevolution among the leaf-cutter agriculture group that exhibit coevolved modifications, to something more like diffuse (‘one-to-many’) coevolution in the (more ancient) lower agriculture groups; with a narrowly diffuse (‘one-to-few’) coevolution in the Cyphomyrmex ant clade (Mehdiabadi et al., 2012)
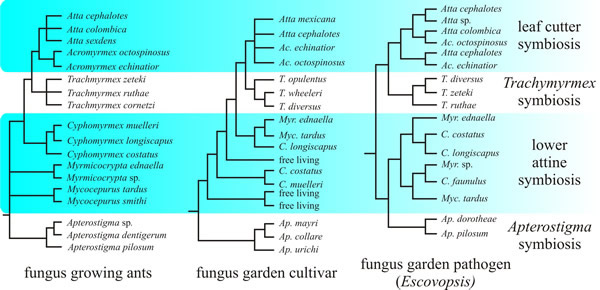 |
Fig. 3. Possible phylogenetic trees of attine ants and their associated fungi. This figure shows the phylogenetic reconstruction of the coevolution of fungus-growing ants (based on the genetic sequences of the ants), their garden fungal cultivars (based on the genetic sequences of the fungi isolated from the gardens), and the fungus garden pathogen Escovopsis (based on the genetic sequences of Escovopsis isolated from the gardens). The different fungus garden cultivar and Escovopsis strains in the cladograms are indicated by the name of the host ant species maintaining the garden from which they were isolated. The similarities in the cladograms of this illustration show the apparent tripartite coevolution of the symbioses, but this may oversimplify a complex Escovopsis population (see text). The highly significant similarity displayed here is that all three phylogenies separate into four major lineages, representing four major evolutionary innovations: (i) the initial lower attine symbiosis between Leucocoprinaceous fungi and the most primitive attine ant species; (ii) the Apterostigma symbiosis, involving Tricholomataceous fungi and a clade of ants derived within the genus Apterostigma; (iii) the symbiosis, incorporating derived Leucocoprinaceous fungi and ants in the genus Trachymyrmex; and (iv) the leaf cutter symbiosis, involving highly derived Leucocoprinaceous fungi and the well-known leaf cutting ant species. Illustration redrawn and modified from Currie et al., 2003. Exact correspondence in branching patterns is not expected for a number of reasons. Overall it seems that attine ants and their fungal partners are sufficiently diverse to represent the full spectrum from ‘one-to-one’ coevolution among the leaf-cutter agriculture group that exhibit coevolved modifications, to something more like diffuse (‘one-to-many’) coevolution in the (more ancient) lower agriculture groups; with a narrowly diffuse (‘one-to-few’) coevolution in the Cyphomyrmex ant clade (Mehdiabadi et al., 2012). |
As Fig. 3 indicates, the association may be even more complex than described so far. There is a common (fungal) parasite of the fungus garden, called Escovopsis, and this too has been claimed to show co-evolution with the ants and their cultivars of fungi in a tripartite evolutionary model (Currie et al., 2003). However, analysis of a much greater range of specimens has found that gardens from different clades of ants can sometimes be infected by closely related strains of Escovopsis, suggesting that the alleged tripartite co-evolution may be spurious.
It seems that Escovopsis shows much greater diversity than previously thought and has a correspondingly more complex evolutionary history (Meirelles et al., 2015). Escovopsis (Ascomycota: anamorphic Hypocreales) is a known parasite of free-living fungi, the ancestors of which were probably associated accidentally with the ancient fungal cultivars. Escovopsis has evolved and has now emerged as a specialised parasite of ant garden fungal cultivars. Interestingly, Escovopsis is a parasite of filamentous hyphae; it does not infect yeast gardens. Avoidance of parasitism by Escovopsis may have been part of the selection pressure favouring yeast growth in the ant-yeast agriculture mutualisms.
The interactions and dependencies go even further than described so far; because yet another organism is involved. The ants have developed a symbiotic relationship with a filamentous actinomycete bacterium of the genus Streptomyces. Areas of the cuticle of fungus-growing ants are coated with what appears to the naked eye to be a powdery, whitish-grey crust which is actually formed from masses of Streptomyces. Actinomycetes are common organisms, mostly soil-dwelling, that produce many secondary metabolites with antibacterial or antifungal properties (most of our own clinical antibiotics are actinomycete metabolites, and many are from the genus Streptomyces).
The Streptomyces associated with fungus-growing ants produces antibiotics that suppress growth of the parasite Escovopsis. This actinomycete is associated with all species of fungus-growing ants so far studied, being carried in genus-specific crypts and exocrine glands on the surface of the ant. The bacteria are transmitted vertically (from parent to offspring colonies), and the association of Streptomyces with attine ants is both highly evolved and of very ancient origin (Currie, 2001; Currie et al., 2003, 2006; Mueller et al, 2001). Effectively, attine ants use multidrug therapy to maintain their fungal cultivars (Barke et al., 2011).
Updated January, 2020
