3.2 The Chytrids
Members of the phylum Chytridiomycota, often referred to as chytrid fungi or chytrids, are morphologically simple organisms with a global distribution and approximately 700 described species that can be found from the tropics to the arctic regions. Chytrids occur in aquatic environments such as streams, ponds, estuaries and marine systems, living as parasites of algae and planktonic organisms. Many chytrids, perhaps the majority, occur in terrestrial forest, agricultural and desert soils, and in acidic bogs as saprotrophs on difficult-to-digest substrata like pollen grains, chitin, keratin and cellulose. Some soil chytrids are obligate parasites of vascular plants. Chytrids now placed in phylum Neocallimastigomycota exist as obligately anaerobic symbionts in the intestines of herbivorous mammals.
Members of the phylum Chytridiomycota, often referred to as chytrid fungi or chytrids, are morphologically simple organisms with a global distribution and approximately 700 described species that can be found from the tropics to the arctic regions. Chytrids occur in aquatic environments such as streams, ponds, estuaries and marine systems, living as parasites of algae and planktonic organisms. Many chytrids, perhaps the majority, occur in terrestrial forest, agricultural and desert soils, and in acidic bogs as saprotrophs on difficult-to-digest substrata like pollen grains, chitin, keratin and cellulose. Some soil chytrids are obligate parasites of vascular plants.
Chytrids reproduce through the production of motile spores (zoospores), which are typically propelled by a single flagellum directed towards the rear. Chytrid morphology is very simple (Fig. 1).
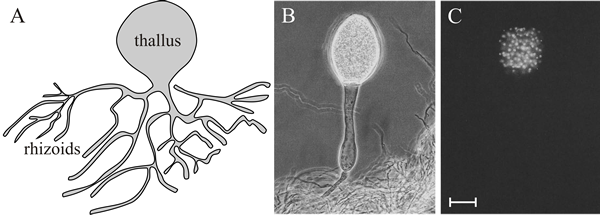 |
Fig. 1. General morphology of chytrids. A, sketch diagram of a eucarpic chytrid thallus. The most obvious morphological feature is the thallus, the main body of the chytrid, perhaps 10 µm diameter, in which most of the cytoplasm resides and from which a system of branching elements emerges. The latter is called the rhizoidal system; the rhizoids anchor the fungus in its substratum and secrete digestive enzymes. The thallus is converted into a sporangium during reproduction, so the sporangium is sac-like and its protoplasm becomes internally divided to produce zoospores. B, phase contrast microphotograph of a thallus of the rumen chytrid, Neocallimastix sp., showing the single sporangium of the monocentric thallus and its rhizoidal system. C, the same field of view as in B, but this time with DAPI (4'-6-diamidino-2-phenylindole) fluorescence staining. DAPI forms fluorescent complexes with natural double-stranded DNA, so very specifically stains nuclei. In image C the fluorescent staining is limited to the thallus/sporangium, showing that the rhizoids do not contain nuclei. Scale bar = 40 µm. Images B and C modified from Trinci et al. 1994. CLICK HERE for more images illustrating chytrids diversity. |
Eucarpic chytrids are those that consist of a sporangium and filamentous rhizoids, and contrast with holocarpic chytrids that produce thalli that are entirely converted to sporangia during reproduction. Zoospore-producing sporangia (zoosporangia; always the result of asexual reproduction) have thin walls. Resting spores may be formed sexually or asexually, they have thick cell walls and may germinate to produce a sporangium after a dormant period.
In monocentric chytrids the thallus produces a single sporangium, whereas those described as polycentric are individual chytrids in which several sporangia form on a network of rhizoids termed a rhizomycelium. Other features used for taxonomy include whether the sporangium has a lid-like operculum that opens to release zoospores, and whether there is (subsporangial) swelling just below the sporangium (called the apophysis). Chytrids have also been distinguished on the basis of whether they grow on (epibiotic) or within (endobiotic) their substratum.
Zoospores are usually 2-10 µm diameter, lack a wall, contain a single nucleus and are propelled by a single posterior whiplash flagellum (though some anaerobic rumen chytrids, closely related but classified in a separate phylum, the Neocallimastigomycota , have multiflagellate zoospores). CLICK HERE to see some illustrations.
Names to look out for among the Chytridiomycota include Rhizophydium, which is the largest genus in the Chytridiales, its members having one reproductive centre per thallus (= monocentric), have rhizoids (= eucarpic) and develop endogenously (the zoospore cyst enlarges to form the zoosporangium). Species of Rhizophydium release zoospores through one or more pores or, in some species, a large part of the sporangial wall melts away to release zoospores. Rhizophydium sphaerotheca develops on pollen grains and Rhizophydium globosum develops on algae. Among plant parasites are Synchytrium on potatoes and Olpidium on curcurbits (including cucumber, squashes, pumpkins, gherkins, melons of all kinds, and various gourds).
The only known chytrid parasites of vertebrates are Batrachochytrium dendrobatidis and B. salamandrivorans which cause a fatal epidermal infection (chytridiomycosis) of amphibian species and has been responsible for mass mortality and major population declines of amphibians around the world (see Section 16.7 Cutaneous chytridiomycosis: an emerging infectious disease of amphibians for more details) ; CLICK HERE to view now). Batrachochytrium grows on the keratinised skin of adult frogs and keratinised mouth parts of tadpoles. The resulting epidermal hyperplasia (increase in the number of the cells as a chronic inflammatory response) impairs cutaneous respiration and osmoregulation causing widespread fatalities in adults. Toxin production has not been implicated.
Although most chytrids are infrequently encountered and scarce to rare, a few species are relatively common, even abundant, in most freshwater and soil habitats (e.g. species of Rhizophydium, Rhizophlyctis, Phlyctochytrium, and Chytriomyces are readily isolated by flooding soil samples with water and ‘baiting’ with cellulose, chitin, pollen grains or hemp seeds). In fact, chytrids are very important components of freshwater ecosystems, and in their review of environmental factors that affect growth and population composition of chytrids in aquatic habitats, Gleason et al. (2008) identify five roles for chytrids in food-web dynamics as:
- chytrid zoospores are a good food source for zooplankton,
- chytrids decompose particulate organic matter,
- chytrids are parasites of aquatic plants,
- chytrids are parasites of aquatic animals
- chytrids convert inorganic compounds into organic compounds.
Anaerobic chytrids are probably the most numerous and the most economically-important members of the group, because they occur in the rumen and hindgut of many larger mammalian herbivores; and that includes all farmed animals (see the section on Neocallimastigomycota (Section 3.3).
There has always been debate about whether chytrids are ‘true’ fungi, but they have chitin in their cell walls, they use the alpha-aminoadipic acid pathway to synthesise lysine and use glycogen as storage carbohydrates; which are all characteristics of true fungi. So, for many years the chytrids have been treated as true fungi, but very primitive ones because of their simple morphology and their dependence on zoospores for sexual reproduction. Recent phylogenetic evidence using mitochondrial genes, nuclear rDNA sequences and whole genomes conclusively demonstrated that chytrids are true fungi and that they occupy a basal position in the fungal phylogenetic tree.
We are basing our descriptions of the group on the AFTOL summary of chytrid phylogeny (James et al., 2006) and the subsequent overall classification of Fungi published by Hibbett et al. (2007). Prior to AFTOL, and on the basis of life cycle and mode of reproduction, together with gross morphology and zoospore ultrastructure, the Chytridiomycota were divided into five orders:
- Chytridiales (characterised by zygotic meiosis in which haploid individuals fuse to form a diploid zygote which immediately undergoes meiosis).
- Spizellomycetales (separated from the Chytridiales on the basis of distinctive ultrastructure).
- Monoblepharidales (which are oogamous).
- Neocallimastigales (anaerobic rumen symbionts).
- Blastocladiales (which have sporic meiosis, in which meiosis results in the production of haploid spores, and an alternation of sporophytic and gametophytic generations).
The AFTOL study identified four major lineages of chytrid fungi (that is, the Chytridiomycota as described above is not monophyletic). The Blastocladiales and Neocallimastigales are both phylogenetically distinct from other chytrids and were elevated to the level of phylum (Blastocladiomycota and Neocallimastigomycota).
Interestingly, the Neocallimastigomycota emerges as the earliest diverging lineage. So, the lineage originated long, long before its current hosts, ruminant herbivores, appeared on the geological scene (see the section entitled Anaerobic fungi and the rise of the ruminants in Chapter 15; CLICK HERE to view now). Removing the Neocallimastigomycota and Blastocladiomycota leaves four Orders within Chytridiomycota: Chytridiales, Spizellomycetales, Rhizophydiales and Monoblepharidales. This summary may need revision as further molecular analyses resolve other phylogenetically distinct clades of flagellated fungi (James et al., 2006; Powell & Letcher, 2014).
Updated September, 2020
